Learning Objectives
After completing this section, you should be able to
-
- write an equation to represent the formation of a soap.
- identify the structure of the fat required to produce a given soap.
- identify the structure of a soap, given the structure of the fat from which it is produced.
- describe the mechanism by which soaps exert their cleansing action.
- give a chemical explanation of the problems encountered when carboxylate soaps are used in hard‑water areas, and explain how they may be overcome by the use of sulphonate detergents.
Key Terms
Make certain that you can define, and use in context, the key terms below.
- hydrophilic
- lipophilic (hydrophobic)
- amphiphilic
- micelles
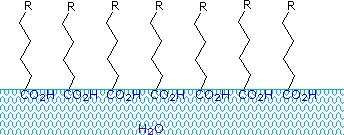
Carboxylic acids and salts having alkyl chains longer than eight carbons exhibit unusual behavior in water due to the presence of both hydrophilic (CO2) and hydrophobic (alkyl) regions in the same molecule. Such molecules are termed amphiphilic (Gk. amphi = both) or amphipathic. Fatty acids made up of ten or more carbon atoms are nearly insoluble in water, and because of their lower density, float on the surface when mixed with water. Unlike paraffin or other alkanes, which tend to puddle on the waters surface, these fatty acids spread evenly over an extended water surface, eventually forming a monomolecular layer in which the polar carboxyl groups are hydrogen bonded at the water interface, and the hydrocarbon chains are aligned together away from the water. This behavior is illustrated in the diagram on the right. Substances that accumulate at water surfaces and change the surface properties are called surfactants.
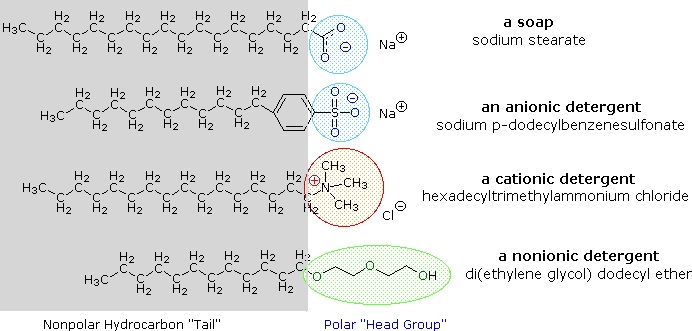
Alkali metal salts of fatty acids are more soluble in water than the acids themselves, and the amphiphilic character of these substances also make them strong surfactants. The most common examples of such compounds are soaps and detergents, four of which are shown below. Note that each of these molecules has a nonpolar hydrocarbon chain, the "tail", and a polar (often ionic) "head group". The use of such compounds as cleaning agents is facilitated by their surfactant character, which lowers the surface tension of water, allowing it to penetrate and wet a variety of materials.
Very small amounts of these surfactants dissolve in water to give a random dispersion of solute molecules. However, when the concentration is increased an interesting change occurs. The surfactant molecules reversibly assemble into polymolecular aggregates called micelles. By gathering the hydrophobic chains together in the center of the micelle, disruption of the hydrogen bonded structure of liquid water is minimized, and the polar head groups extend into the surrounding water where they participate in hydrogen bonding. These micelles are often spherical in shape, but may also assume cylindrical and branched forms, as illustrated on the right. Here the polar head group is designated by a blue circle, and the nonpolar tail is a zig-zag black line.
The oldest amphiphilic cleaning agent known to humans is soap. Soap is manufactured by the base-catalyzed hydrolysis (saponification) of animal fat (see below). Before sodium hydroxide was commercially available, a boiling solution of potassium carbonate leached from wood ashes was used. Soft potassium soaps were then converted to the harder sodium soaps by washing with salt solution. The importance of soap to human civilization is documented by history, but some problems associated with its use have been recognized. One of these is caused by the weak acidity (pKa ca. 4.9) of the fatty acids. Solutions of alkali metal soaps are slightly alkaline (pH 8 to 9) due to hydrolysis. If the pH of a soap solution is lowered by acidic contaminants, insoluble fatty acids precipitate and form a scum. A second problem is caused by the presence of calcium and magnesium salts in the water supply (hard water). These divalent cations cause aggregation of the micelles, which then deposit as a dirty scum.
These problems have been alleviated by the development of synthetic amphiphiles called detergents (or syndets). By using a much stronger acid for the polar head group, water solutions of the amphiphile are less sensitive to pH changes. Also the sulfonate functions used for virtually all anionic detergents confer greater solubility on micelles incorporating the alkaline earth cations found in hard water. Variations on the amphiphile theme have led to the development of other classes, such as the cationic and nonionic detergents shown above. Cationic detergents often exhibit germicidal properties, and their ability to change surface pH has made them useful as fabric softeners and hair conditioners. These versatile chemical "tools" have dramatically transformed the household and personal care cleaning product markets over the past fifty years
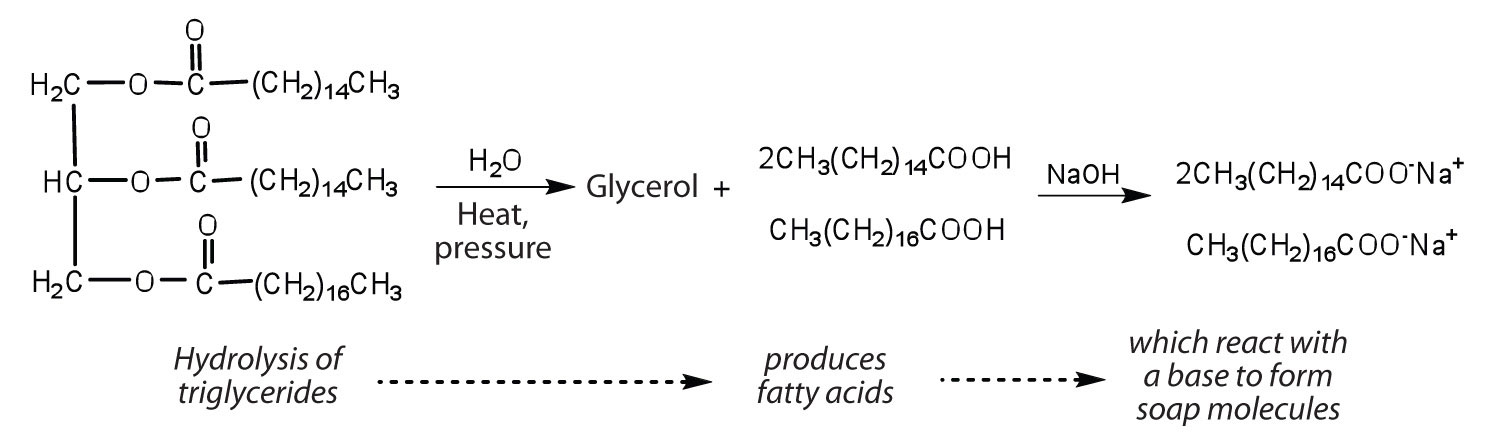
Chemical Reactions of Fats and Oils
Fats and oils can participate in a variety of chemical reactions—for example, because triglycerides are esters, they can be hydrolyzed in the presence of an acid, a base, or specific enzymes known as lipases. The hydrolysis of fats and oils in the presence of a base is used to make soap and is called saponification. Today most soaps are prepared through the hydrolysis of triglycerides (often from tallow, coconut oil, or both) using water under high pressure and temperature [700 lb/in2 (∼50 atm or 5,000 kPa) and 200°C]. Sodium carbonate or sodium hydroxide is then used to convert the fatty acids to their sodium salts (soap molecules):
Contributors and Attributions