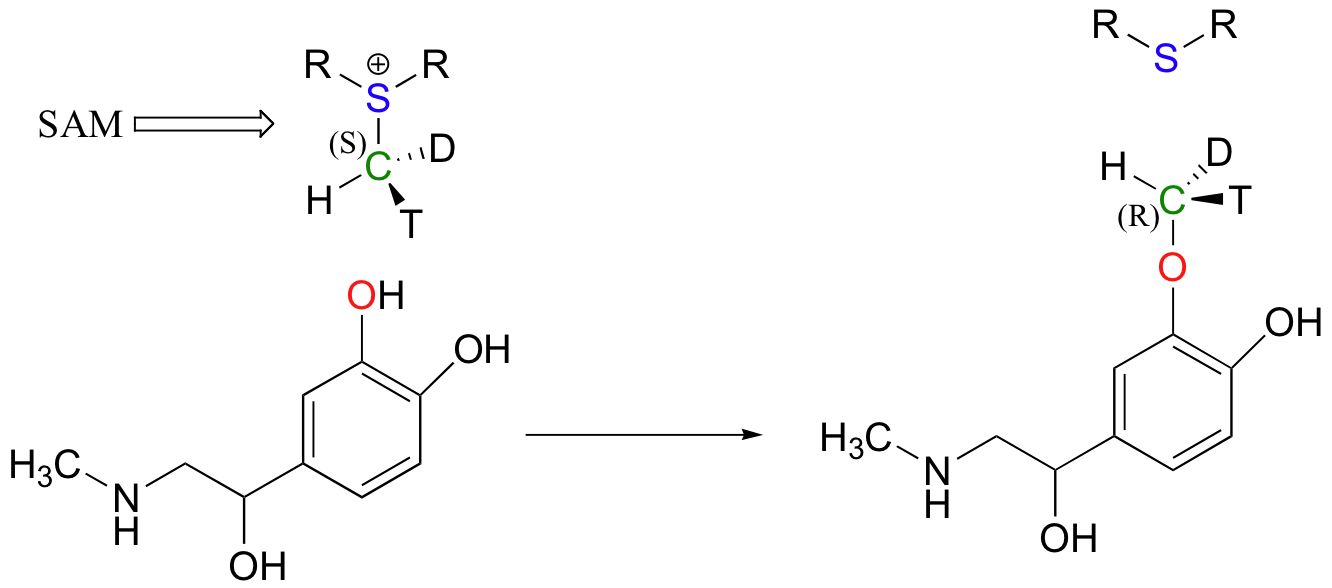Leaving Groups in Biochemical Reactions
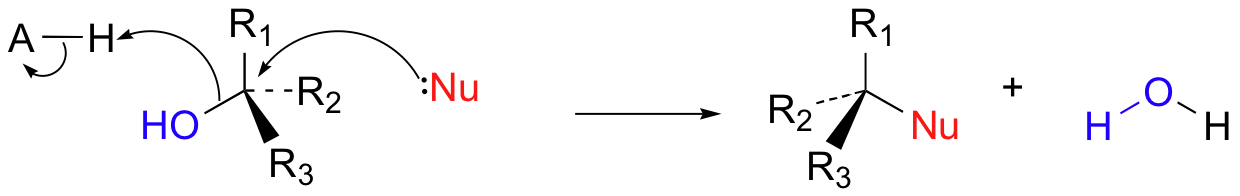
In biological reactions, we do not often see halides serving as leaving groups (in fact, outside of some marine organisms, halogens are fairly unusual in biological molecules). More common leaving groups in biochemical reactions are phosphates, water, alcohols, and thiols. In many cases, the leaving group is protonated by an acidic group on the enzyme as bond-breaking occurs. For example, hydroxide ion itself seldom acts as a leaving group – it is simply too high in energy (too basic). Rather, the hydroxide oxygen is generally protonated by an enzymatic acid before or during the bond-breaking event, resulting in a (very stable) water leaving group.
More often, however, the hydroxyl group of an alcohol is first converted enzymatically to a phosphate ester in order to create a better leaving group. This phosphate ester can take the form of a simple monophosphate (arrow 1 in the figure below), a diphosphate (arrow 2), or a nucleotide monophosphate (arrow 3).
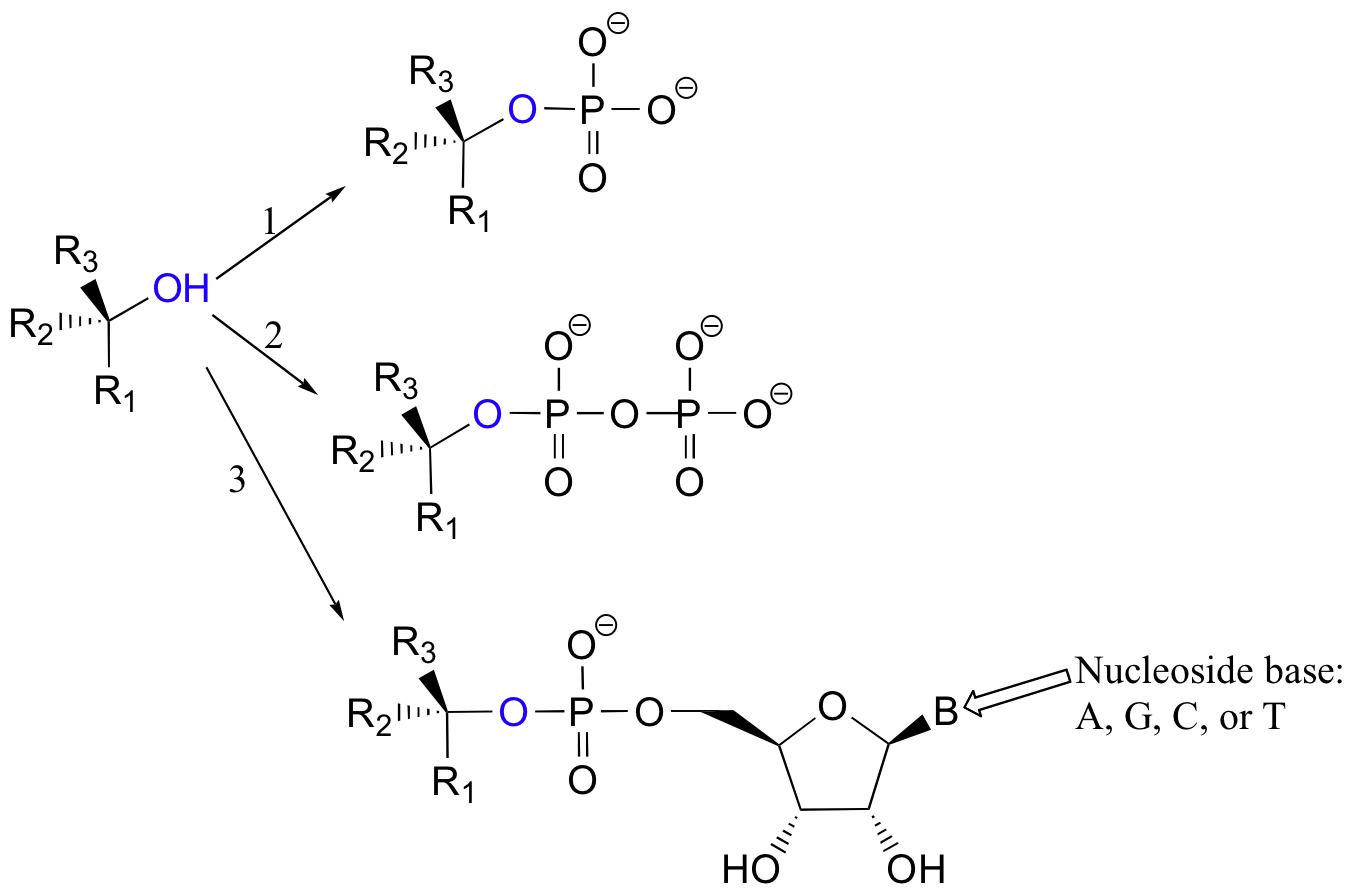
Due to resonance delocalization of the developing negative charge, phosphates are excellent leaving groups.

Here’s a specific example (from DNA nucleotide biosynthesis):
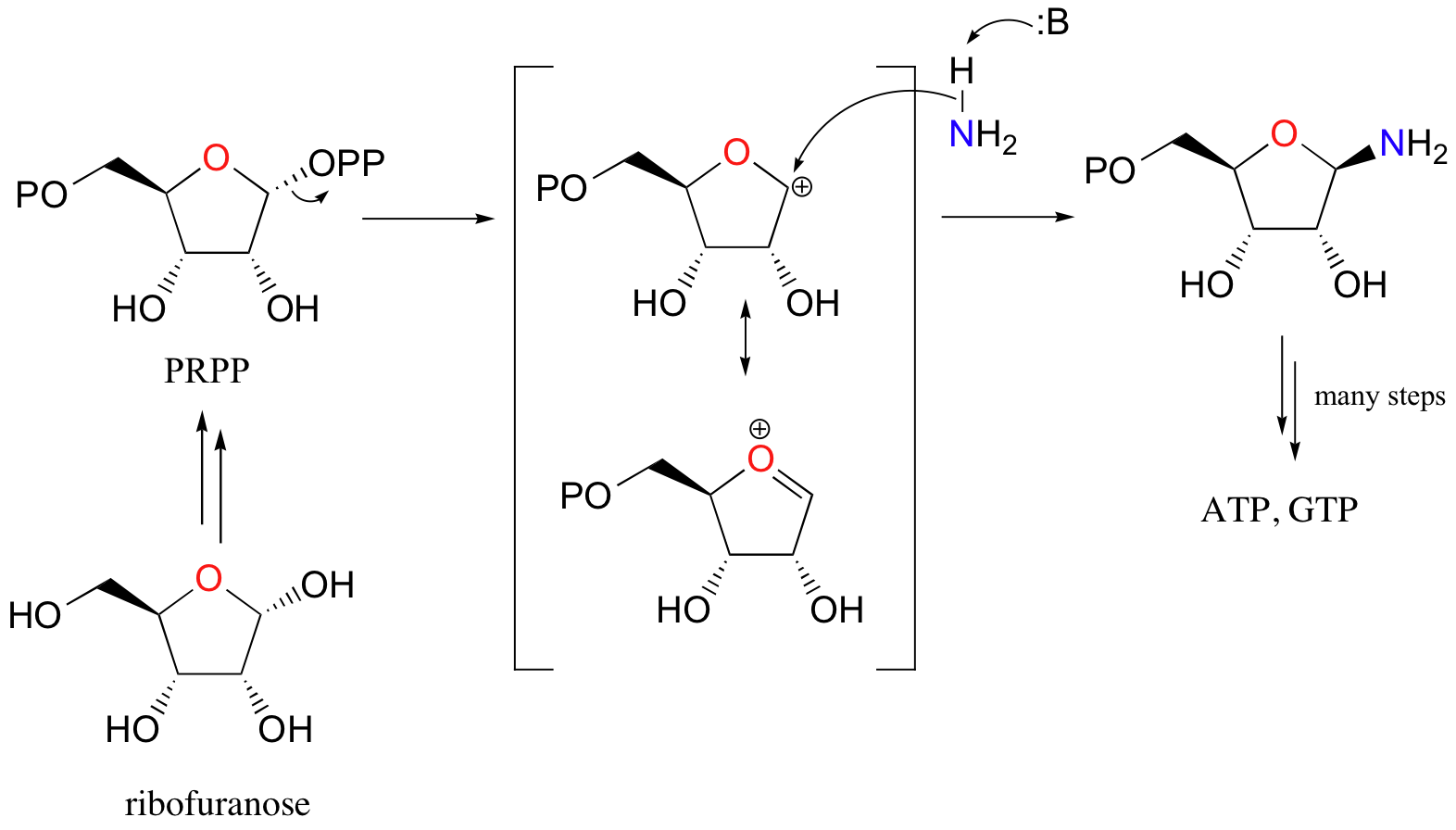
Here, the OH group on ribofuranose is converted to a diphosphate, a much better leaving group. Ammonia is the nucleophile in the second step of this SN1-like reaction.
What is important for now is that in each case, an alcohol has been converted into a much better leaving group, and is now primed for a nucleophilic substitution reaction.
SAM Methyltransferases
Some of the most important examples of SN2 reactions in biochemistry are those catalyzed by S-adenosyl methionine (SAM) – dependent methyltransferase enzymes. We have already seen, in chapter 6 and again in chapter 8, how a methyl group is transferred in an SN2 reaction from SAM to the amine group on the nucleotide base adenosine:
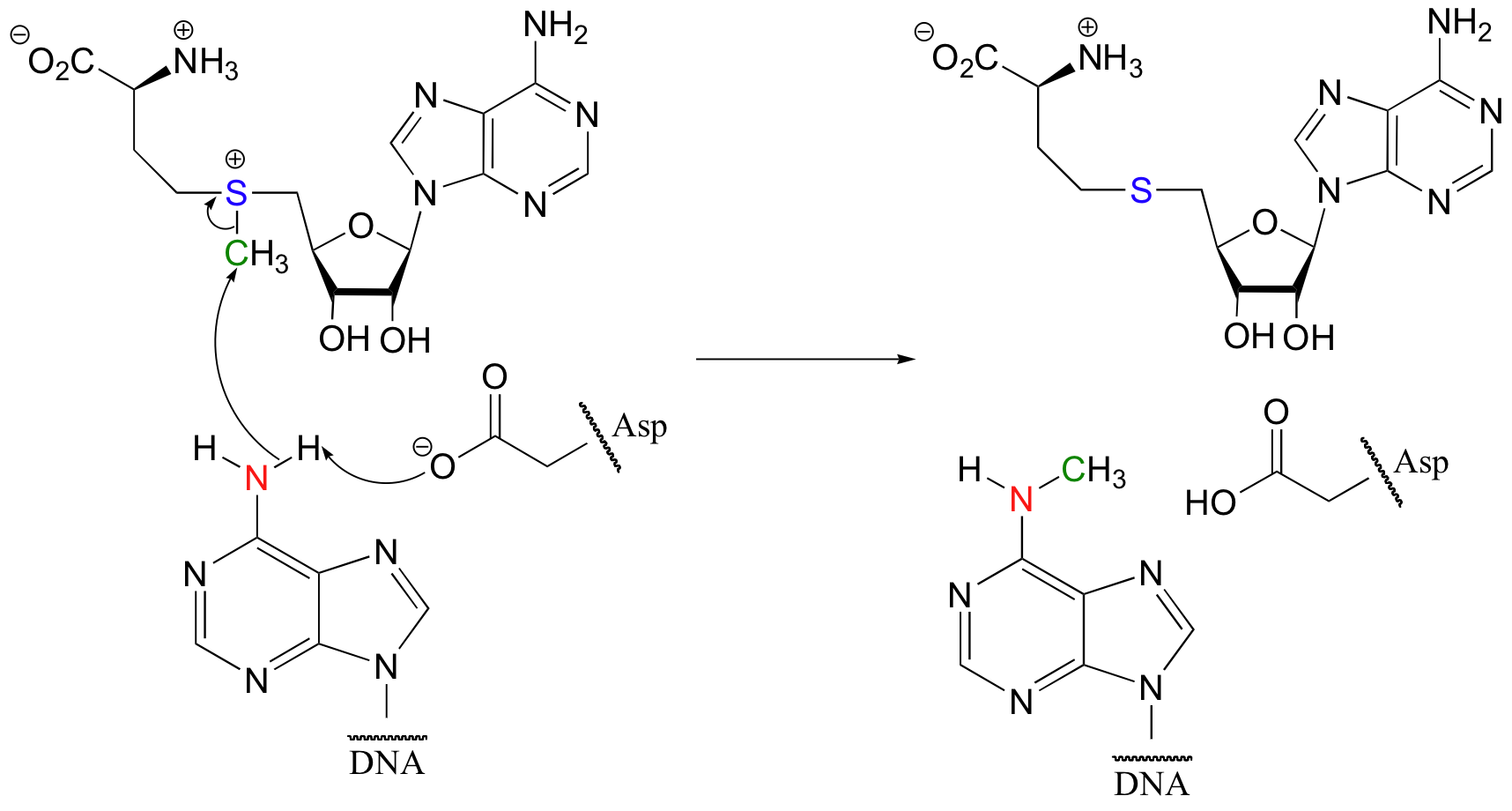
(Nucleic Acids Res. 2000, 28, 3950).
Another SAM-dependent methylation reaction is catalyzed by an enzyme called catechol-O-methyltransferase. The substrate here is epinephrine, also known as adrenaline.
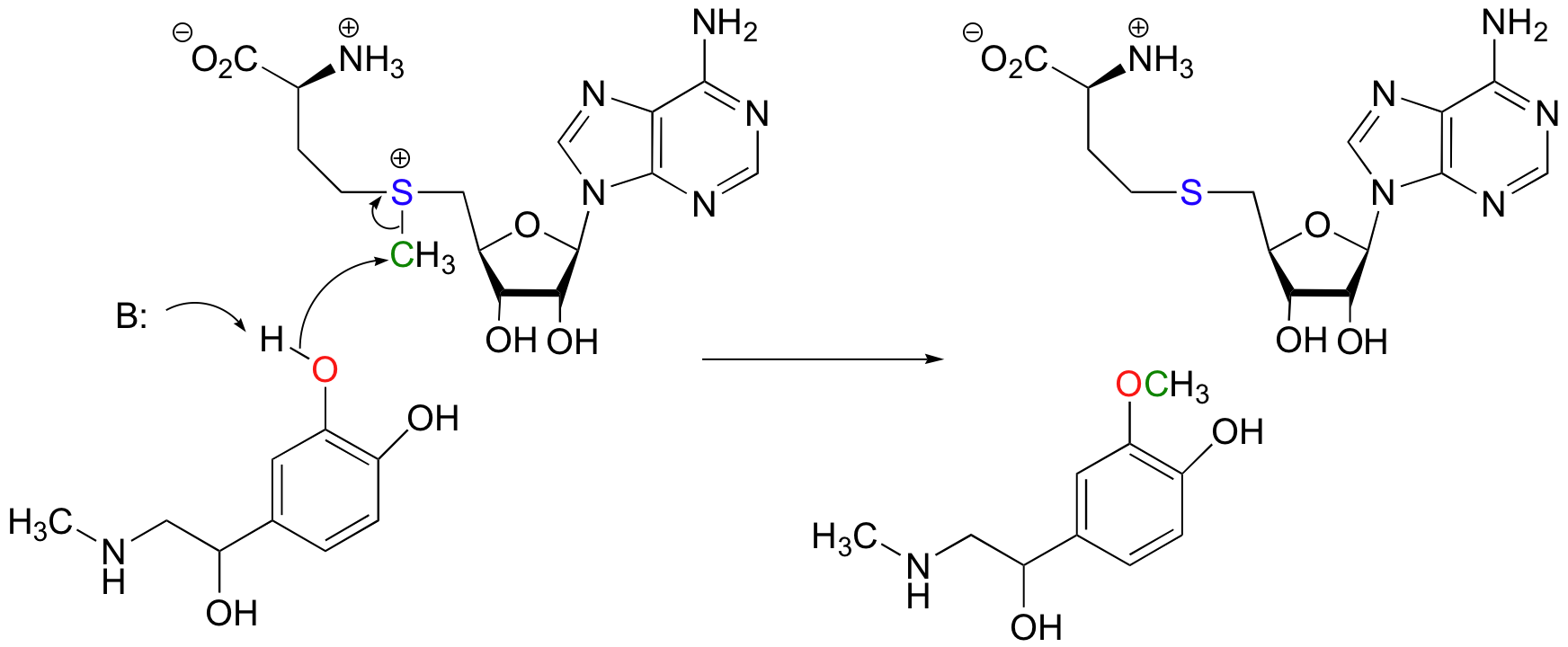
Notice that in this example, the attacking nucleophile is an alcohol rather than an amine (that’s why the enzyme is called an O-methyltransferase). In both cases, though, a basic amino acid side chain is positioned in the active site in just the right place to deprotonate the nucleophilic group as it attacks, increasing its nucleophilicity. The electrophile in both reactions is a methyl carbon, so there is little steric hindrance to slow down the nucleophilic attack. The methyl carbon is electrophilic because it is bonded to a positively-charged sulfur, which is a powerful electron withdrawing group. The positive charge on the sulfur also makes it an excellent leaving group, as the resulting product will be a neutral and very stable sulfide. All in all, in both reactions we have a reasonably good nucleophile, an electron-poor, unhindered electrophile, and an excellent leaving group.
Because the electrophilic carbon in these reactions is a methyl carbon, a stepwise SN1-like mechanism is extremely unlikely: a methyl carbocation is very high in energy and thus is not a reasonable intermediate to propose. We can confidently predict that this reaction is SN2. Does this SN2 reaction occur, as expected, with inversion of stereochemistry? Of course, the electrophilic methyl carbon in these reactions is achiral, so inversion is not apparent. To demonstrate inversion, the following experiment has been carried out with catechol-O-methyltransferase:
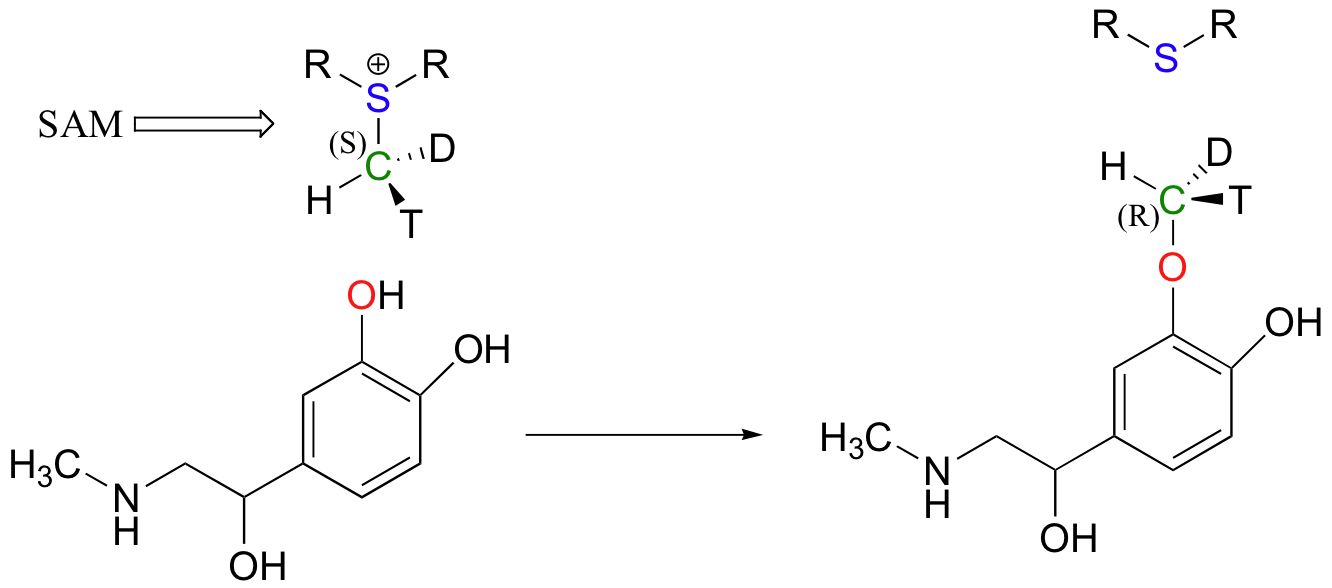
Here, the methyl group of SAM was made to be chiral by incorporating hydrogen isotopes tritium (3H, T) and deuterium (2H, D). The researchers determined that the reaction occurred with inversion of configuration, as expected for an SN2 displacement (J. Biol. Chem. 1980, 255, 9124).