The high reactivity of free radicals and the multiplicative nature of radical chain reactions can be useful in the synthesis of materials such as polyethylene plastic - but these same factors can also result in dangerous consequences in a biological or ecological context. You are probably aware of the danger posed to the earth's protective stratospheric ozone layer by the use of chlorofluorocarbons (\(CFCs\)) as refrigerants and propellants in aerosol spray cans. Freon-11, or \(CFCl_3\), is a typical \(CFC\) that was widely used until late in the 20th century. It can take months or years for a \(CFC\) molecule to drift up into the stratosphere from the surface of the earth, and of course the concentration of \(CFCs\) at this altitude is very low. Ozone, on the other hand, is continually being formed in the stratosphere. Why all the concern, then, about destruction of the ozone layer - how could such a small amount of \(CFCs\) possibly do significant damage? The problem lies in the fact that the process by which ozone is destroyed is a chain reaction, so that a single \(CFC\) molecule can initiate the destruction of many ozone molecules before a chain termination event occurs.
Although there are several different processes by which the ozone destruction process might occur, the most important is believed to be the chain reaction shown below.
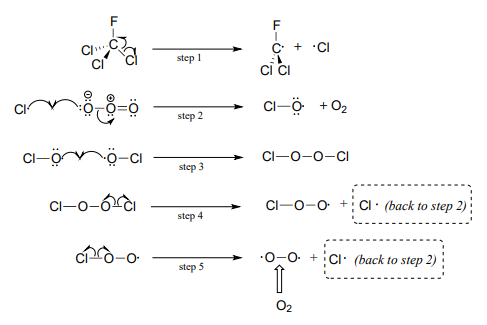
First, a \(CFC\) molecule undergoes homolytic cleavage upon exposure to UV radiation, resulting in the formation of two radicals (step 1). The chlorine radical rapidly reacts with ozone (step 2) to form molecular oxygen and a chlorine monoxide radical. Step 3 appears to be a chain termination step, as two chlorine monoxide radicals combine. The \(Cl_2O_2\) condensation product, however, is highly reactive and undergoes two successive homolytic cleavage events (steps 4 and 5) to form \(O_2\) and two chlorine radicals, which propagates the chain.
To address the problem of ozone destruction, materials chemists have developed new hydrofluorocarbon refrigerant compounds. The newer compounds contain carbon-hydrogen bonds, which are weaker than the carbon-halogen bonds in \(CFCs\), and thus are susceptible to homolytic cleavage caused by small amounts of hydroxide radical present in the lower atmosphere:
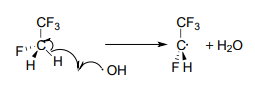
This degradation occurs before the refrigerant molecules have a chance to drift higher up to the stratosphere where the ozone plays its important protective role. The degradation products are quite unstable and quickly degrade further into relatively harmless by-products. The hydroxide radical is sometimes referred to as an atmospheric 'detergent' due to its ability to degrade escaped refrigerants and other volatile organic pollutants.
Hydrofluorocarbons do, however, act as greenhouse gases, and are thought to contribute to climate change.




