Learning Objectives
- To classify a given chemical reaction into a variety of types.
Although there are untold millions of possible chemical reactions, most can be classified into a small number of general reaction types. Classifying reactions has two purposes: it helps us to recognize similarities among them, and it enables us to predict the products of certain reactions. A particular reaction may fall into more than one of the categories that we will define in this book.
Combination (composition) Reactions
A combination (composition) reaction is a chemical reaction that makes a single substance from two or more reactants. There may be more than one molecule of product in the balanced chemical equation, but there is only one substance produced. For example, the equation
\[\ce{4Fe + 3O_2 \rightarrow 2Fe_2O_3} \nonumber \]
is a combination reaction that produces \(\ce{Fe2O3}\) from its constituent elements — \(\ce{Fe}\) and \(\ce{O2}\). Combination reactions do not have to combine elements, however. The chemical equation
\[\ce{Fe_2O_3 + 3SO_3 \rightarrow Fe_2(SO_4)_3} \nonumber \]
shows a combination reaction in which \(\ce{Fe2O3}\) combines with three molecules of \(\ce{SO3}\) to make \(\ce{Fe2(SO4)3}\).
Example \(\PageIndex{1}\)
Which equations are combination reactions?
- \(\ce{Co(s) + Cl2(g) → CoCl2(s)}\)
- \(\ce{CO(g) + Cl2(g) → COCl2(g)}\)
- \(\ce{N2H4(ℓ) + O2(g) → N2(g) + 2H2O(ℓ)}\)
Solution
- This is a combination reaction.
- This is a combination reaction. (The compound \(\ce{COCl2}\) is called phosgene and, in the past, was used as a gassing agent in chemical warfare.)
- This is not a combination reaction.
Exercise \(\PageIndex{1}\)
Which equations are combination reactions?
- \(\ce{P4(s) + 6Cl2(g) → 4PCl3(g)}\)
- \(\ce{SO3(ℓ) + H2O(ℓ) → H2SO4(ℓ)}\)
- \(\ce{NaOH(s) + HCl(g) → NaCl(s) + H2O(ℓ)}\)
- Answer
-
a and b are combination reactions
Decomposition Reactions
A decomposition reaction is the reverse of a combination reaction. In a decomposition reaction, a single substance is converted into two or more products. There may be more than one molecule of the reactant, but there is only one substance initially. For example, the equation
\[\ce{2NaHCO3(s) \rightarrow Na_2CO3(s) + CO2(g) + H_2O (ℓ) } \nonumber \]
is a decomposition reaction that occurs when \(\ce{NaHCO3}\) is exposed to heat. Another example is the decomposition of \(\ce{KClO3}\):
\[\ce{2KClO3 (s) \rightarrow 2KCl (s) + 3O2 (g)} \nonumber \]
This reaction was once commonly used to generate small amounts of oxygen in the chemistry lab.
The decomposition reaction of \(\ce{NaHCO3}\) is the reaction that occurs when baking soda is poured on a small kitchen fire. The intent is that the \(\ce{H2O}\) and \(\ce{CO2}\) produced by the decomposition will smother the flames.
Combustion Reactions
A combustion reaction occurs when a substance combines with molecular oxygen to make oxygen-containing compounds of other elements in the reaction. Many combustion reactions occur with a hydrocarbon, a compound made up solely of carbon and hydrogen. The products of the combustion of hydrocarbons are carbon dioxide and water. Many hydrocarbons are used as fuel because their combustion releases very large amounts of heat energy. An example is propane (\(\ce{C3H8}\)), a gaseous hydrocarbon that is commonly used as the fuel source in gas grills.
\[\ce{C3H8(g) + 5O2(g) → 3CO2(g) + 4H2O(g)} \nonumber\]
Another example is the burning of acetylene (\(\ce{C2H2}\)) in welding torches:
\[\ce{2C2H2 + 5O2 \rightarrow 4CO2 + 2H2O } \nonumber \]
Oxygen (in its elemental form) is a crucial reactant in combustion reactions.
Energy in the form of heat is usually given off as a product in a combustion reaction as well.
Example \(\PageIndex{2}\)
Carbon dioxide (\(\ce{CO2}\)) is an important heat-trapping (greenhouse) gas, which is released through human activities such as deforestation and burning fossil fuels, as well as natural processes such as respiration and volcanic eruptions. The graph above shows \(\ce{CO2}\) levels during the last three glacial cycles, as reconstructed from ice cores.
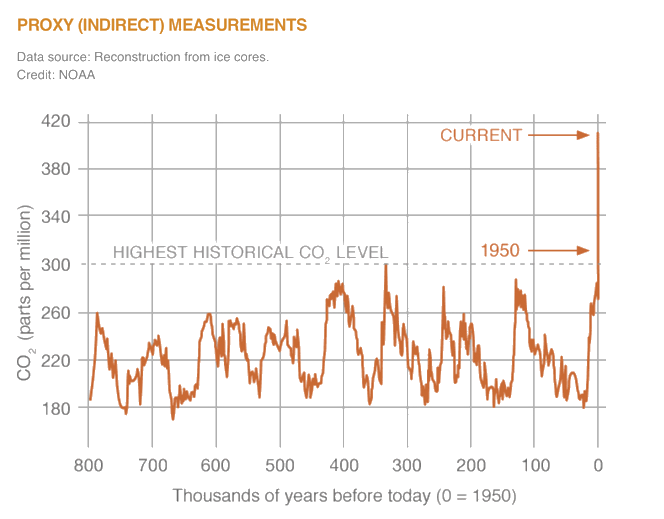
Carbon dioxide is the primary greenhouse gas emitted through human activities. In 2015, \(\ce{CO2}\) accounted for about 82.2% of all U.S. greenhouse gas emissions from human activities. Carbon dioxide is naturally present in the atmosphere as part of the Earth's carbon cycle (the natural circulation of carbon among the atmosphere, oceans, soil, plants, and animals). Human activities are altering the carbon cycle–both by adding more \(\ce{CO2}\) to the atmosphere and by influencing the ability of natural sinks, like forests, to remove \(\ce{CO2}\) from the atmosphere. While \(\ce{CO2}\) emissions come from a variety of natural sources, human-related emissions are responsible for the increase that has occurred in the atmosphere since the industrial revolution.
The main human activity that emits \(\ce{CO2}\) is the combustion of fossil fuels (coal, natural gas, and oil) for energy and transportation, although certain industrial processes and land-use changes also emit \(\ce{CO2}\). An example of how \(\ce{CO2}\) can be generated is the combustion of octane (\(\ce{C8H10}\)), a component of gasoline.
Write the balanced equation to represent the combustion of octane.
Solution
\[\ce{2C8H10(l) + 25O2(g) → 16CO2(g) + 18H2O(g)} \nonumber\]
The balanced reaction argues that for every two molecules of octane that are burned, 16 molecules of \(\ce{CO2}\) are generated.
Example \(\PageIndex{2}\)
Identify each type of reaction.
- \(\ce{2K(s) + S(s) + 2O2(g) → K2SO4(s)}\)
- \(\ce{(NH4)2Cr2O7(s) → N2(g) + Cr2O3(s) + 4H2O(ℓ)}\)
- \(\ce{CH4(g) + 2O2(g) → CO2(g) + 2H2O(ℓ)}\)
Solution
- Multiple reactants are combining to make a single product, so this reaction is a combination reaction.
- A single substance reacts to make several products, so we have a decomposition reaction.
- Oxygen reacts with a compound to make carbon dioxide (an oxide of carbon) and water (an oxide of hydrogen). This is a combustion reaction.
Exercise \(\PageIndex{2}\)
Identify each type of reaction.
- \(\ce{C2H5OH + 3O2 → 2CO2 + 3H2O}\)
- \(\ce{2Mg(s) + O2(g) → 2MgO(s)}\)
- \(\ce{CaCO3(s) → CaO(s) + CO2(g)}\)
- Answer a
-
combustion
- Answer b
-
combination (also combustion)
- Answer c
-
decomposition
Key Takeaway
There are several recognizable types of chemical reactions: combination, decomposition, and combustion reactions are examples.


