20.1: Prelude to Chemical Equilibria
- Page ID
- 238267
The small is great, the great is small; all is in equilibrium in necessity... - Victor Hugo in “Les Miserables”
As was discussed in Chapter 6, the natural tendency of chemical systems is to seek a state of minimum Gibbs function. Once the minimum is achieved, movement in any chemical direction will not be spontaneous. It is at this point that the system achieves a state of equilibrium.
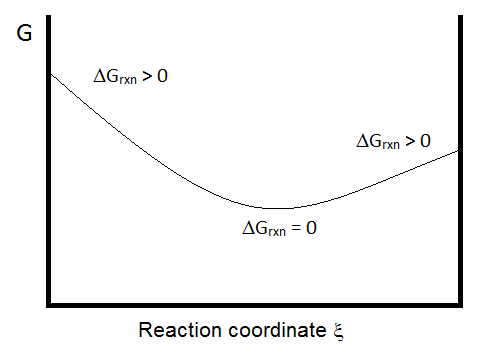
From the diagram above, it should be clear that the direction of spontaneous change is determined by minimizing
\[\left(\frac{\partial G}{\partial \xi}\right)_{P,T}.\]
If the slope of the curve is negative, the reaction will favor a shift toward products. And if it is positive, the reaction will favor a shift toward reactants. This is a non-trivial point, as it underscores the importance of the composition of the reaction mixture in the determination of the direction of the reaction.
Contributors
Patrick E. Fleming (Department of Chemistry and Biochemistry; California State University, East Bay)


