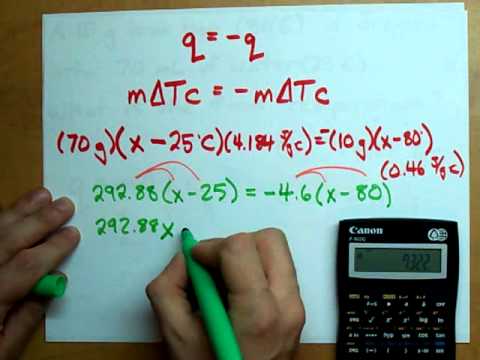9.6: Videos- Calorimetry
- Page ID
- 233050
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \) \( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)\(\newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\) \( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\) \( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\) \( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\) \( \newcommand{\Span}{\mathrm{span}}\) \(\newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\) \( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\) \( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\) \( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\) \( \newcommand{\Span}{\mathrm{span}}\)\(\newcommand{\AA}{\unicode[.8,0]{x212B}}\)
Use the formula mΔTc = –mΔTc to show that heat gained = heat lost and solve for whatever “x” is. In this case, it’s the final temperature.
Calorimetry: Crash Course Chemistry #19
Today’s episode dives into the how of enthalpy. How we calculate it, and how we determine it experimentally . . . even if our determinations here at Crash Course Chemistry are somewhat shoddy.
All rights reserved content
- Calorimetry: Using q=mu0394Tc to find Temperature + Example. Authored by: chemistNATE. Located at: https://youtu.be/llsBOI7BORI. License: All Rights Reserved. License Terms: Standard YouTube License
- Chemistry I: Heat Capacity and Bomb Calorimetry (Medium Lvl Question). Authored by: OneClass. Located at: https://youtu.be/JtvBjmFNy5c. License: All Rights Reserved. License Terms: Standard YouTube License
- Calorimetry: Crash Course Chemistry #19. Authored by: CrashCourse. Located at: https://youtu.be/JuWtBR-rDQk. License: All Rights Reserved. License Terms: Standard YouTube License