1.1: The Scope of Chemistry
- Last updated
- Save as PDF
- Page ID
- 118769
Learning Objectives
- To recognize the breadth, depth, and scope of chemistry.
- Define chemistry in relation to other sciences.
- Identify the main disciplines of chemistry.
Chemistry is the study of matter—what it consists of, what its properties are, and how it changes. Matter is anything that has mass and takes up space—that is, anything that is physically real. Some things are easily identified as matter—the screen on which you are reading this book, for example. Others are not so obvious. Because we move so easily through air, we sometimes forget that it, too, is matter. Because of this, chemistry is a science that has its fingers in just about everything. Being able to describe the ingredients in a cake and how they change when the cake is baked, for example, is chemistry!
Chemistry is one branch of science. Science is the process by which we learn about the natural universe by observing, testing, and then generating models that explain our observations. Because the physical universe is so vast, there are many different branches of science (Figure \(\PageIndex{1}\)). Thus, chemistry is the study of matter, biology is the study of living things, and geology is the study of rocks and the earth. Mathematics is the language of science, and we will use it to communicate some of the ideas of chemistry.
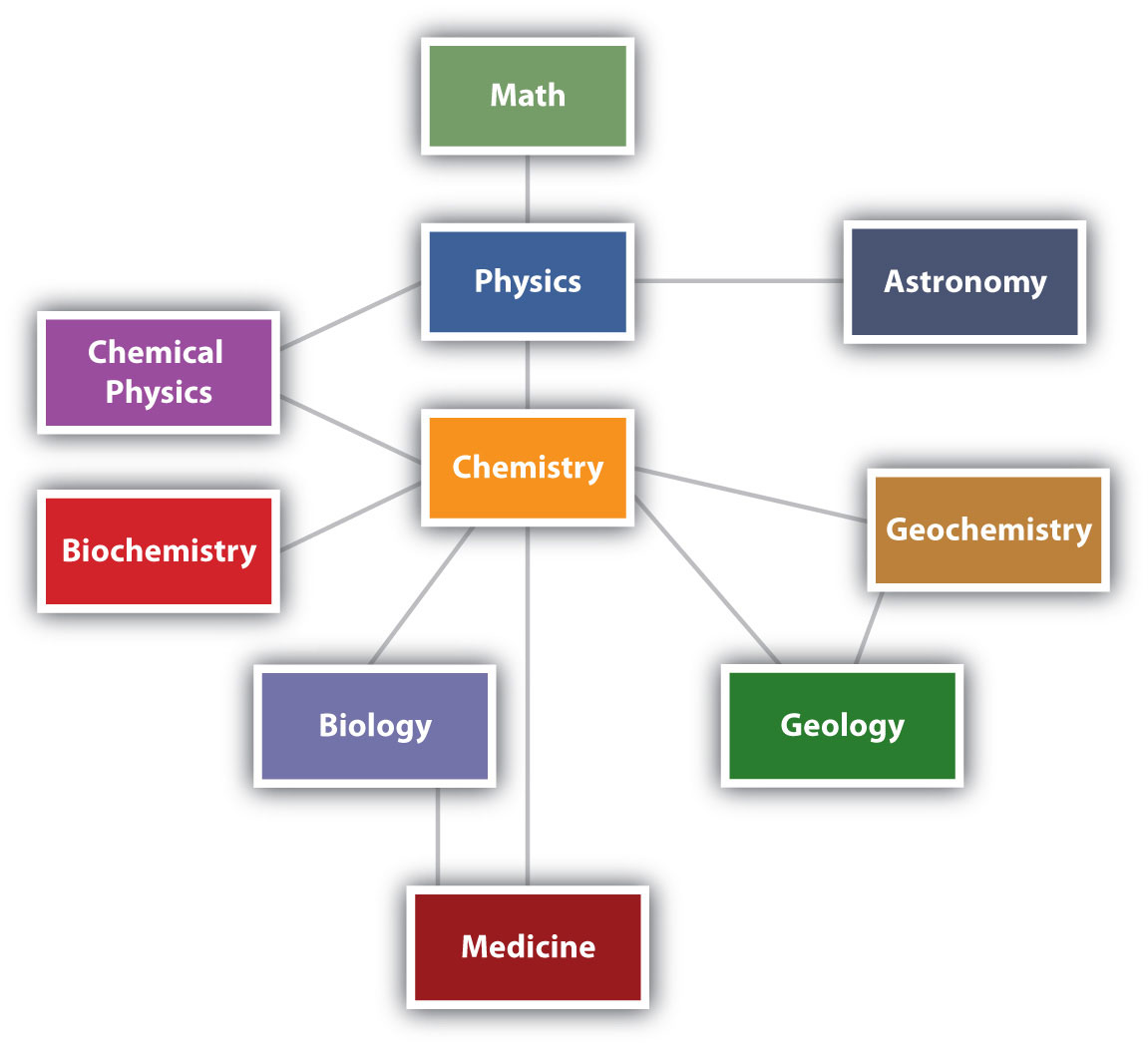
Although we divide science into different fields, there is much overlap among them. For example, some biologists and chemists work in both fields so much that their work is called biochemistry. Similarly, geology and chemistry overlap in the field called geochemistry. Figure \(\PageIndex{1}\) shows how many of the individual fields of science are related. At some level, all of these fields depend on matter because they all involve "stuff"; because of this, chemistry has been called the "central science", linking them all together.
There are many other fields of science, in addition to the ones (biology, medicine, etc.) listed here.
Example \(\PageIndex{1}\): Science Fields
Which fields of study are branches of science? Explain.
- sculpture
- astronomy
Solution
- Sculpture is not considered a science because it is not a study of some aspect of the natural universe.
- Astronomy is the study of stars and planets, which are part of the natural universe. Astronomy is therefore a field of science.
Exercise \(\PageIndex{1}\)
Which fields of study are branches of science?
- physiology (the study of the function of an animal’s or a plant’s body)
- geophysics
- agriculture
- politics
- Answer a:
- yes
- Answer b:
- yes
- Answer c:
- yes
- Answer d:
- no
Areas of Chemistry
The study of modern chemistry has many branches, but can generally be broken down into five main disciplines, or areas of study:
- Physical chemistry: Physical chemistry is the study of macroscopic properties, atomic properties, and phenomena in chemical systems. A physical chemist may study such things as the rates of chemical reactions, the energy transfers that occur in reactions, or the physical structure of materials at the molecular level.
- Organic chemistry: Organic chemistry is the study of chemicals containing carbon. Carbon is one of the most abundant elements on Earth and is capable of forming a tremendously vast number of chemicals (over twenty million so far). Most of the chemicals found in all living organisms are based on carbon.
- Inorganic chemistry: Inorganic chemistry is the study of chemicals that, in general, are not primarily based on carbon. Inorganic chemicals are commonly found in rocks and minerals. One current important area of inorganic chemistry deals with the design and properties of materials involved in energy and information technology.
- Analytical chemistry: Analytical chemistry is the study of the composition of matter. It focuses on separating, identifying, and quantifying chemicals in samples of matter. An analytical chemist may use complex instruments to analyze an unknown material in order to determine its various components.
- Biochemistry: Biochemistry is the study of chemical processes that occur in living things. Research may cover anything from basic cellular processes up to understanding disease states so that better treatments can be developed.


In practice, chemical research is often not limited to just one of the five major disciplines. A particular chemist may use biochemistry to isolate a particular chemical found in the human body such as hemoglobin, the oxygen carrying component of red blood cells. He or she may then proceed to analyze the hemoglobin using methods that would pertain to the areas of physical or analytical chemistry. Many chemists specialize in areas that are combinations of the main disciplines, such as bioinorganic chemistry or physical organic chemistry.
History of Chemistry
The history of chemistry is an interesting and challenging one. Very early chemists were often motivated mainly by the achievement of a specific goal or product. Making perfume or soaps did not need a lot of theory, just a good recipe and careful attention to detail. There was no standard way of naming materials (and no periodic table that we could all agree on). It is often difficult to figure out exactly what a particular person was using. However, the science developed over the centuries by trial and error.
Major progress was made toward putting chemistry on a solid foundation when Robert Boyle (1637-1691) began his research in chemistry (Figure \(\PageIndex{3}\)). He developed the basic ideas about the behavior of gases. He could then describe gases mathematically. Boyle also helped form the idea that small particles could combine to form molecules. Many years later, John Dalton used these ideas to develop the atomic theory.

The field of chemistry began to develop rapidly in the 1700's. Joseph Priestley (1733-1804) isolated and characterized several gases: oxygen, carbon monoxide, and nitrous oxide. It was later discovered that nitrous oxide ("laughing gas") worked as an anesthetic. This gas was used for that purpose for the first time in 1844 during a tooth extraction. Other gases discovered during that time were chlorine, by C.W. Scheele (1742-1786) and nitrogen, by Antoine Lavoisier (1743-1794). Lavoisier has been considered by many scholars to be the "father of chemistry". Among other accomplishments, he discovered the role of oxygen in combustion and definitively formulated the law of conservation of matter.
Chemists continued to discover new compounds in the 1800's. The science also began to develop a more theoretical foundation. John Dalton (1766-1844) put forth his atomic theory in 1807. This idea allowed scientists to think about chemistry in a much more systematic way. Amadeo Avogadro (1776-1856) laid the groundwork for a more quantitative approach to chemistry by calculating the number of particles in a given amount of a gas. A lot of effort was put forth in studying chemical reactions. These efforts led to new materials being produced. Following the invention of the battery by Alessandro Volta (1745-1827), the field of electrochemistry (both theoretical and applications) developed through major contributions by Humphry Davy (1778-1829) and Michael Faraday (1791-1867). Other areas of the discipline also progressed rapidly.
It would take a large book to cover developments in chemistry during the twentieth century and up to today. One major area of expansion was in the area of the chemistry of living processes. Research in photosynthesis in plants, the discovery and characterization of enzymes as biochemical catalysts, elucidation of the structures of biomolecules such as insulin and DNA—these efforts gave rise to an explosion of information in the field of biochemistry.
The practical aspects of chemistry were not ignored. The work of Volta, Davy, and Faraday eventually led to the development of batteries that provided a source of electricity to power a number of devices (Figure \(\PageIndex{4}\)).

Charles Goodyear (1800-1860) discovered the process of vulcanization, allowing a stable rubber product to be produced for the tires of all the vehicles we have today. Louis Pasteur (1822-1895) pioneered the use of heat sterilization to eliminate unwanted microorganisms in wine and milk. Alfred Nobel (1833-1896) invented dynamite (Figure \(\PageIndex{5}\)). After his death, the fortune he made from this product was used to fund the Nobel Prizes in science and the humanities. J.W. Hyatt (1837-1920) developed the first plastic. Leo Baekeland (1863-1944) developed the first synthetic resin, widely used for inexpensive and sturdy dinnerware.

Today, chemistry continues to be essential to the development of new materials and technologies, from semiconductors for electronics to powerful new medicines, and beyond.
Summary
- Chemistry is the study of matter and the changes it undergoes and considers both macroscopic and microscopic information.
- Matter is anything that has mass and occupies space.
- The five main disciplines of chemistry are physical chemistry, organic chemistry, inorganic chemistry, analytical chemistry and biochemistry.
- Many civilizations contributed to the growth of chemistry. A lot of early chemical research focused on practical uses. Basic chemistry theories were developed during the nineteenth century. New materials and batteries are a few of the products of modern chemistry.

