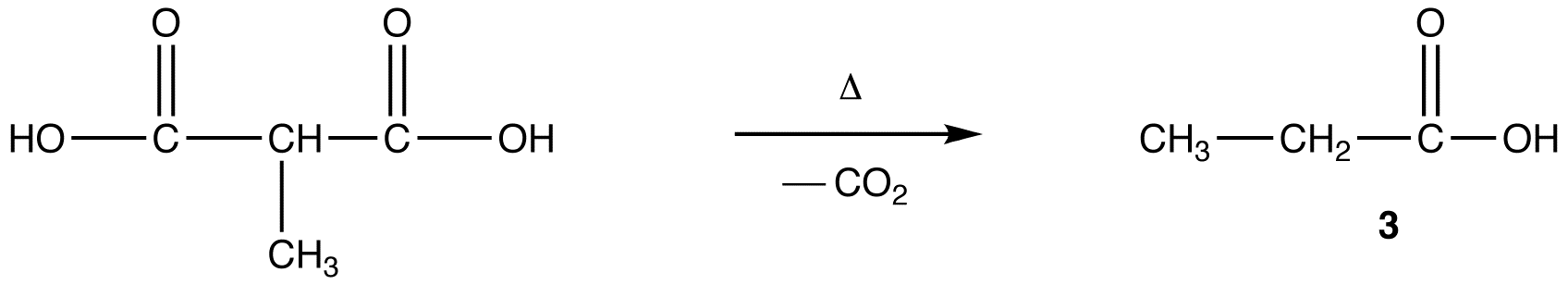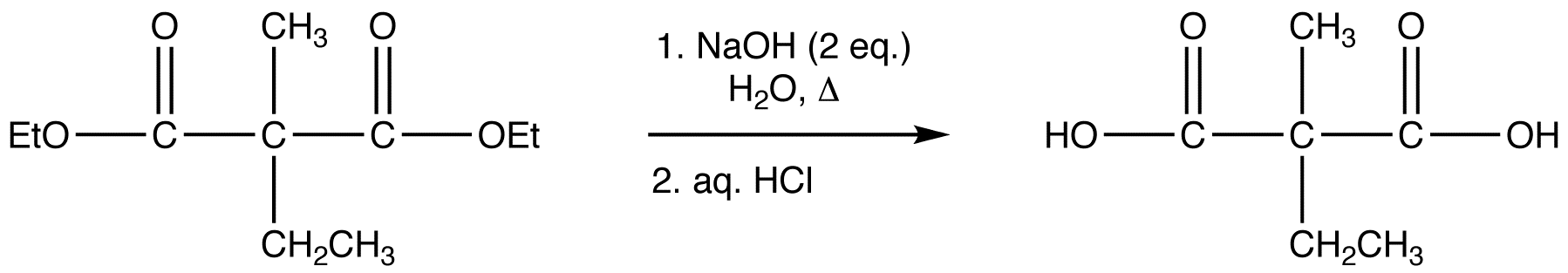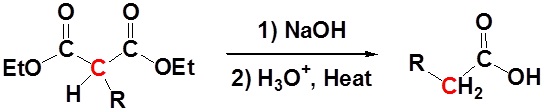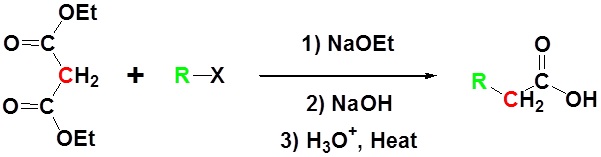Enolates can act as a nucleophile in SN2 type reactions. Overall an α hydrogen is replaced with an alkyl group. This reaction is one of the more important for enolates because a carbon-carbon bond is formed. These alkylations are affected by the same limitations as SN2 reactions previously discussed. A good leaving group, Chloride, Bromide, Iodide, Tosylate, should be used. Also, secondary and tertiary leaving groups should not be used because of poor reactivity and possible competition with elimination reactions. Lastly, it is important to use a strong base, such as LDA or sodium amide, for this reaction. Using a weaker base such as hydroxide or an alkoxide leaves the possibility of multiple alkylation’s occurring.

| Example 1: Alpha Alkylation |
| 
|
Mechanism
1) Enolate formation

2) Sn2 attack

Alkylation of Unsymmetrical Ketones
Unsymmetrical ketones can be regioselctively alkylated to form one major product depending on the reagents.
Treatment with LDA in THF at -78oC tends to form the less substituted kinetic enolate.

Using sodium ethoxide in ethanol at room temperature forms the more substituted thermodynamic enolate.

Problems
1) Please write the structure of the product for the following reactions.

Answers
1)

Malonic ester is a reagent specifically used in a reaction which converts alkyl halides to carboxylic acids called the Malonic Ester Synthesis. Malonic ester synthesis is a synthetic procedure used to convert a compound that has the general structural formula 1 into a carboxylic acid that has the general structural formula 2.

- R1 = alkyl group
- L = leaving group
The group —CH2CO2H in 2 is contributed by a malonic ester, hence the term malonic ester synthesis.
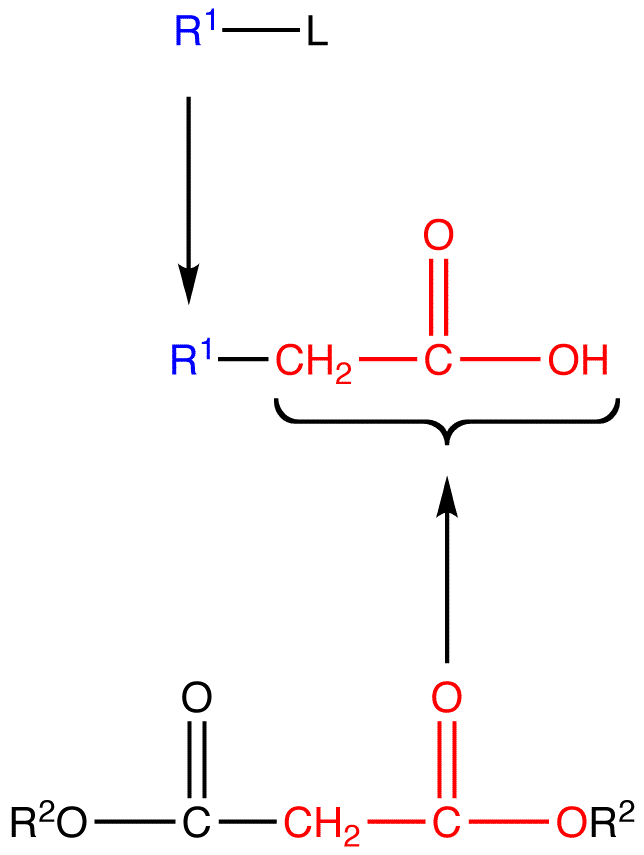
Mechanism
Malonic ester synthesis consists of four consecutive reactions that can be carried out in the same pot.
- reaction 1: acid-base reaction
- reaction 2: nucleophilic substitution
- reaction 3: ester hydrolysis (using saponification)
- reaction 4: decarboxylation
eg:
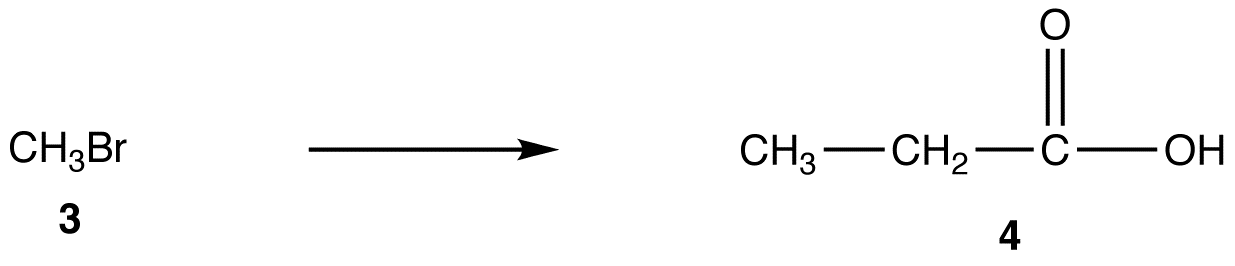
reaction 1:

reaction 2:
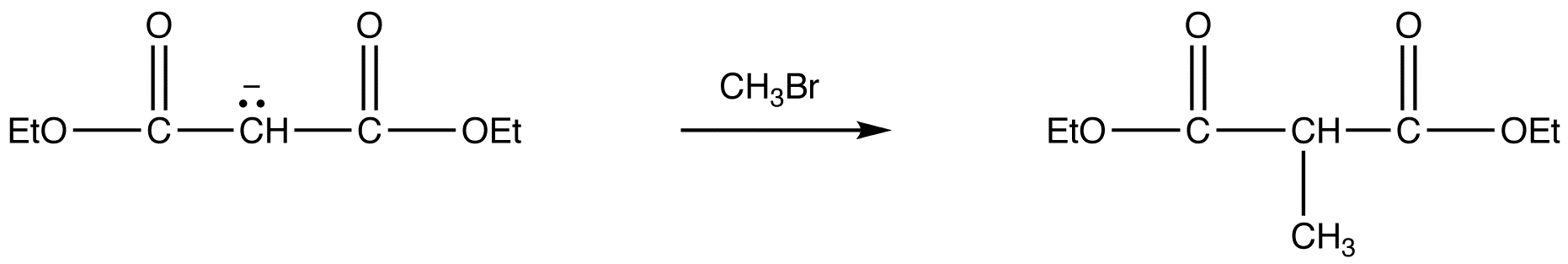
reaction 3:
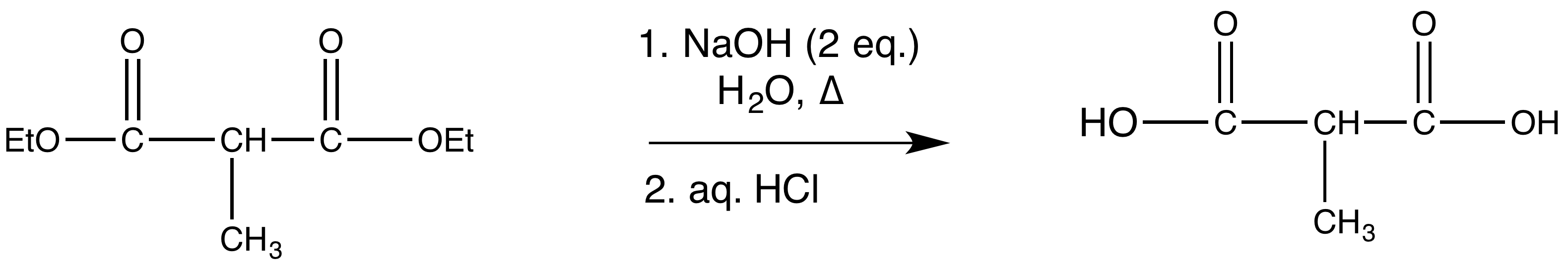
reaction 4:
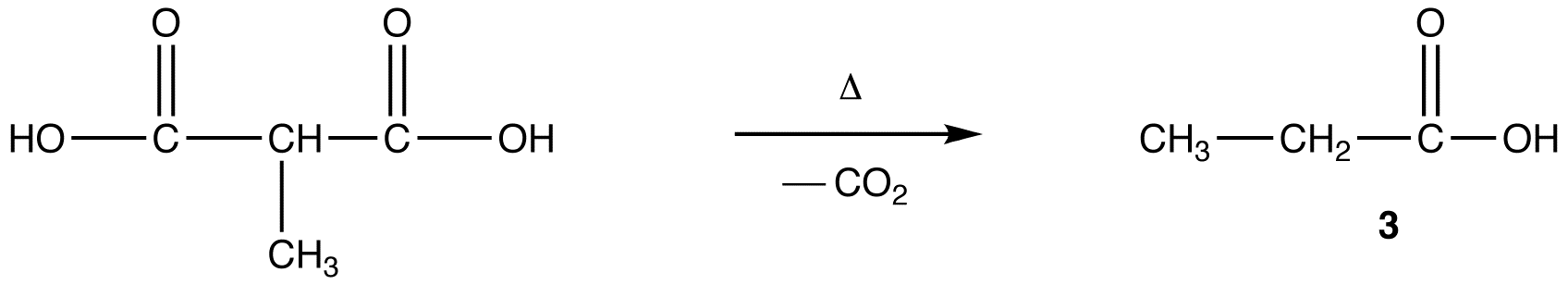
A more direct method to convert 3 into 4 is the reaction of 3 with the enolate ion (5) of ethyl acetate followed by hydrolysis of the resultant ester.
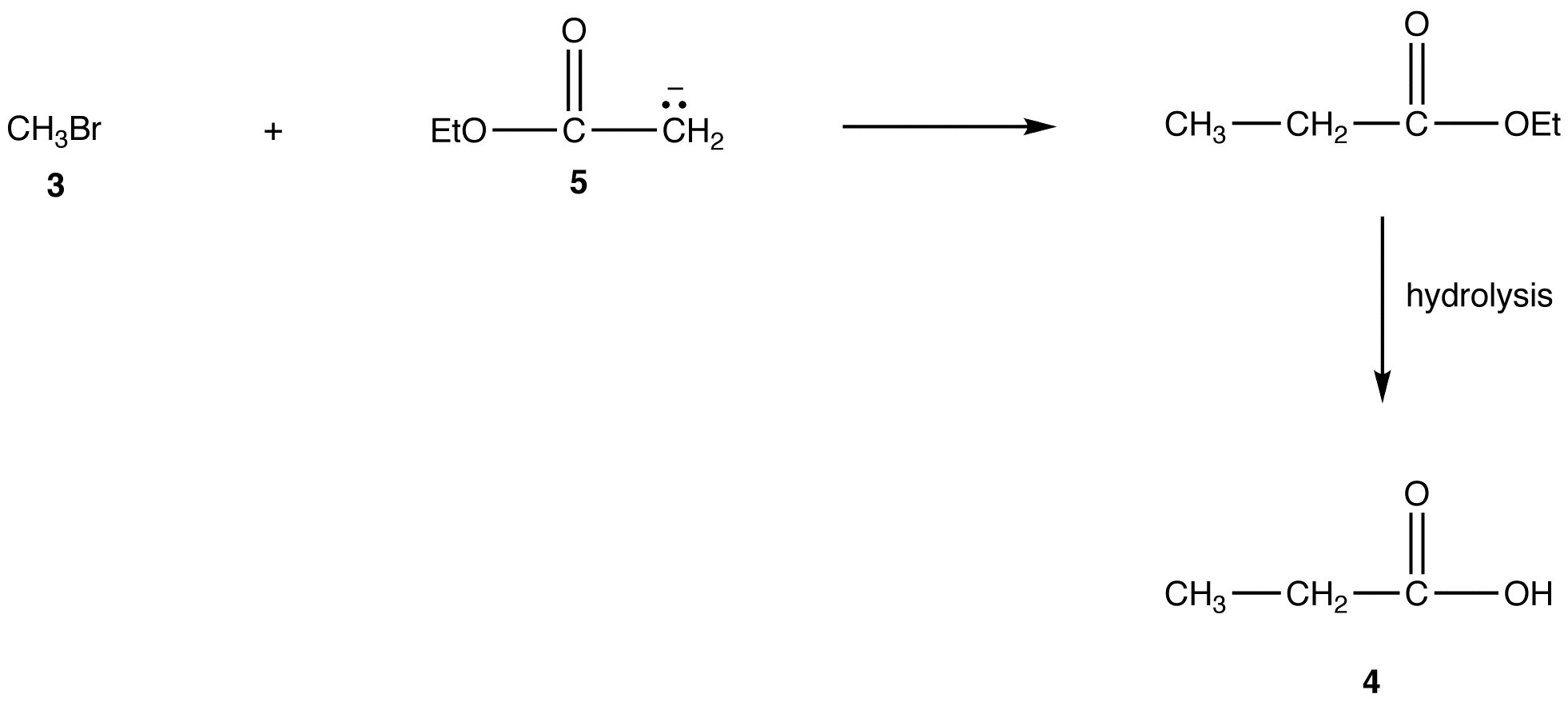
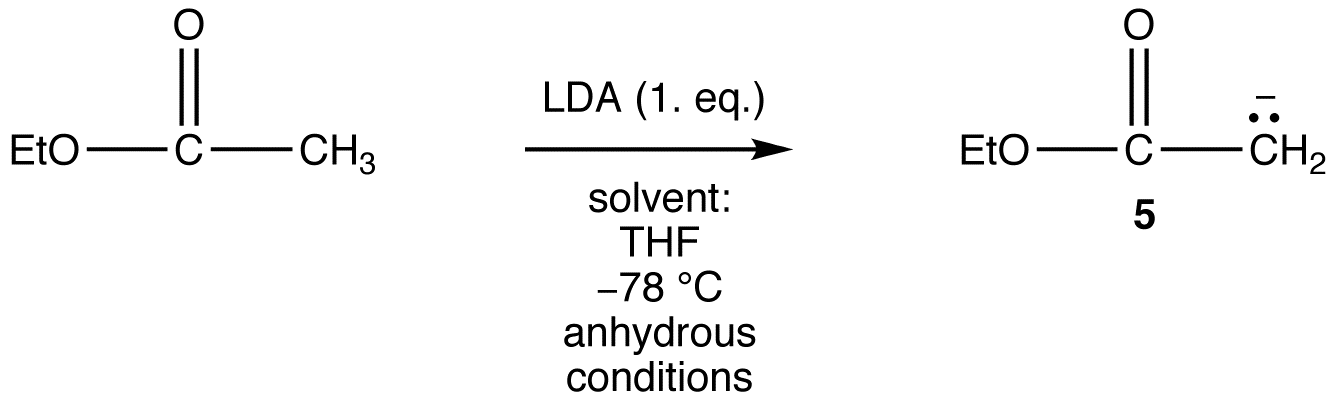
However, the generation of 5 from ethyl acetate quantitatively in high yield is not an easy task because the reaction requires a very strong base, such as LDA, and must be carried out at very low temperature under strictly anhydrous conditions.
Malonic ester synthesis provides a more convenient alternative to convert 3 to 4.
Malonic ester synthesis can be adapted to synthesize compounds that have the general structural formula 6.

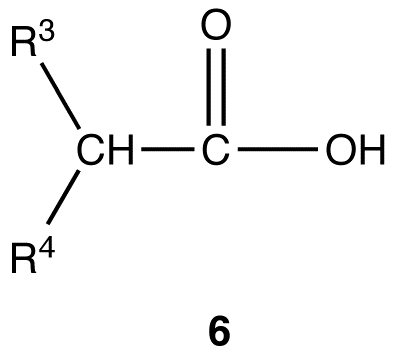
R3, R4 = identical or different alkyl groups
eg:
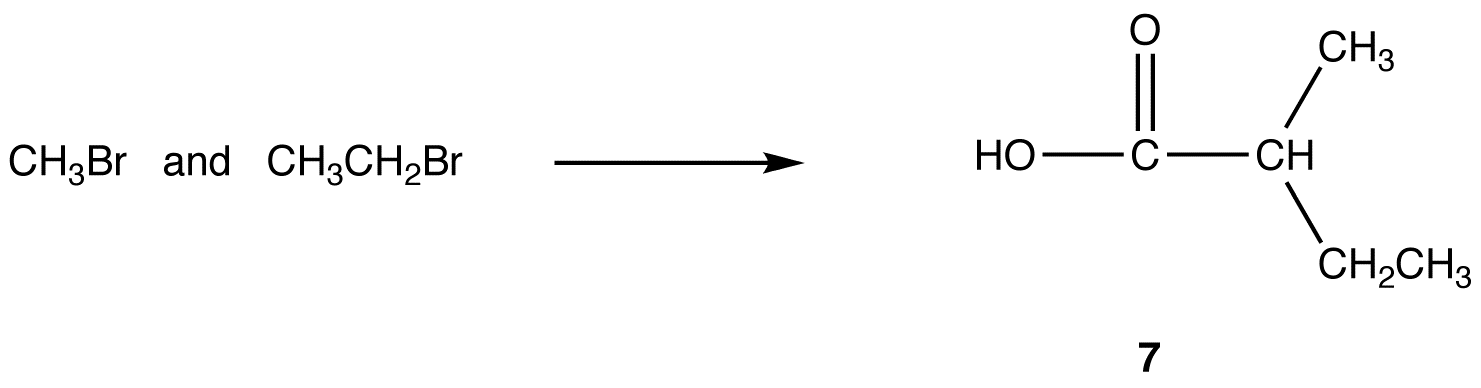
reaction 1:
reaction 2:
reaction 1 (repeat):
reaction 2 (repeat):
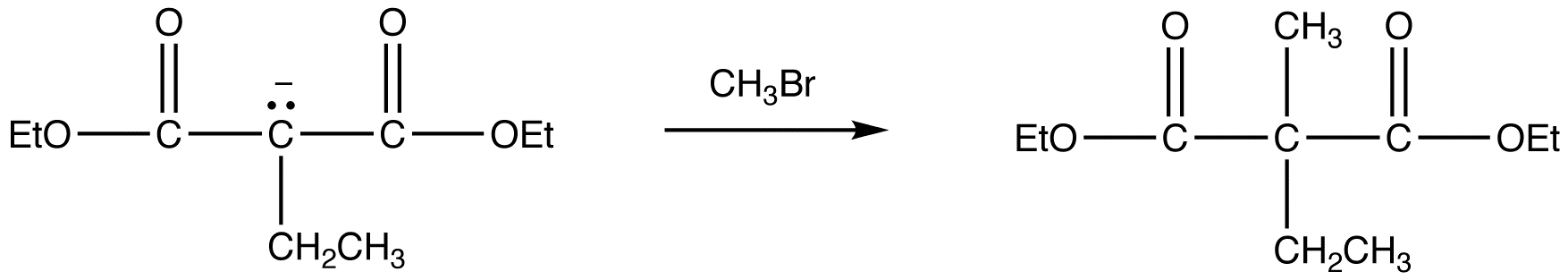
reaction 3:
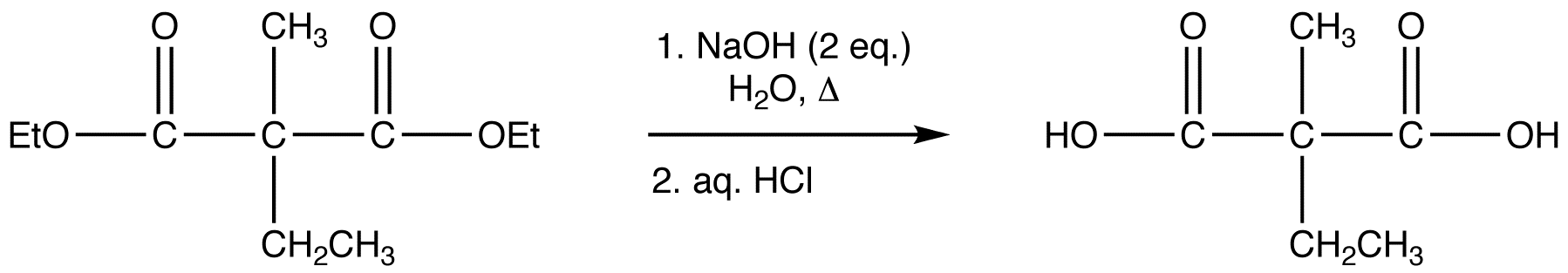
reaction 4:
Malonic Ester Synthesis

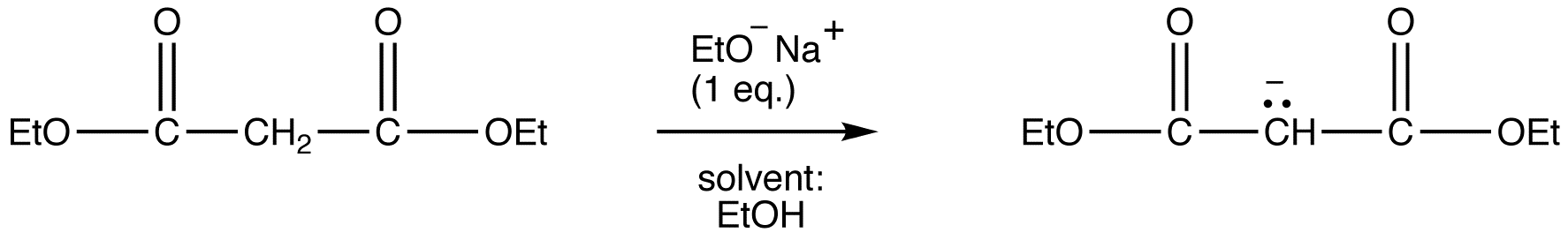
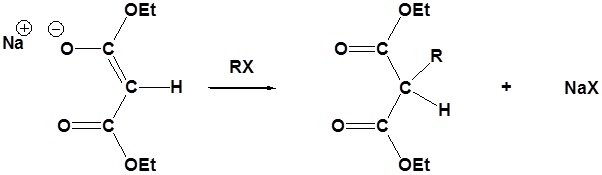
Due to the fact that Malonic ester’s α hydrogens are adjacent to two carbonyls, they can be deprotonated by sodium ethoxide (NaOEt) to form Sodio Malonic Ester.

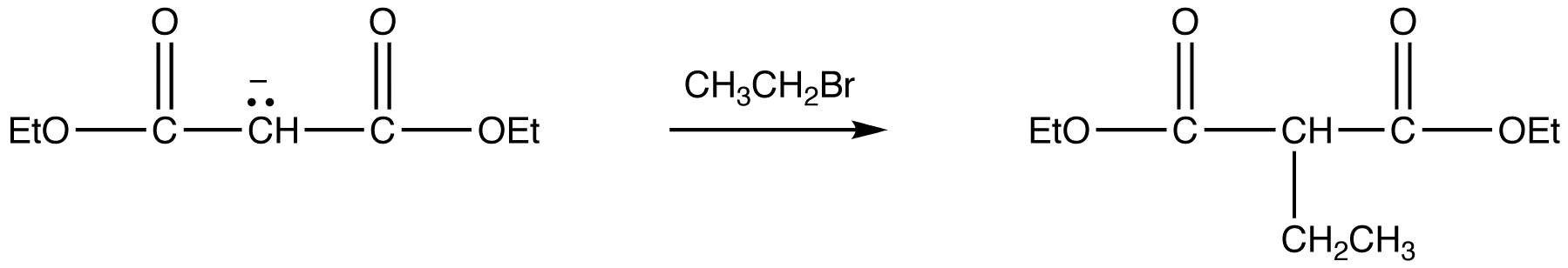
Because Sodio Malonic Ester is an enolate, it can then be alkylated with alkyl halides.
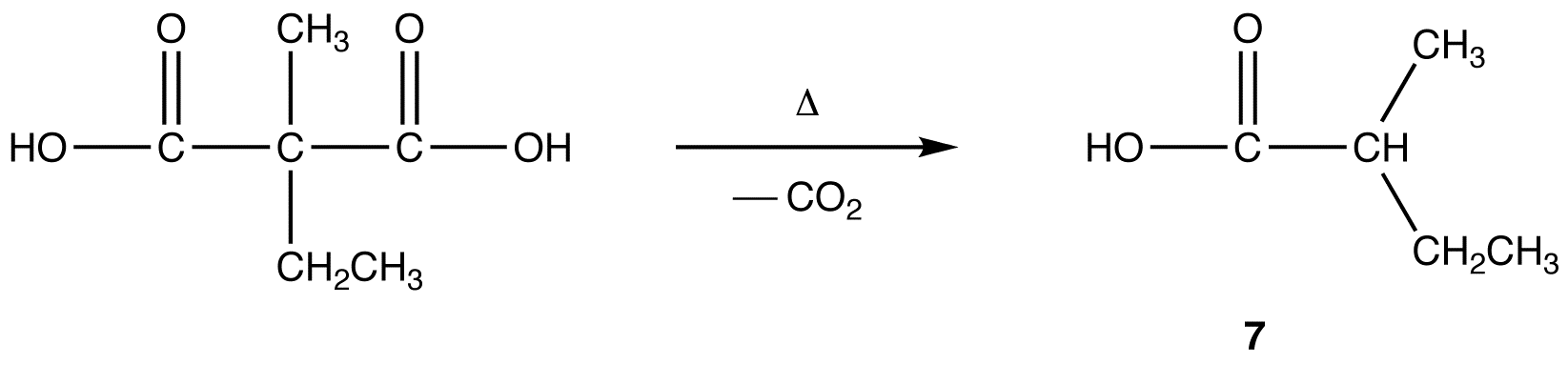
After alkylation the product can be converted to a dicarboxylic acid through saponification and subsequently one of the carboxylic acids can be removed through a decarboxylation step.
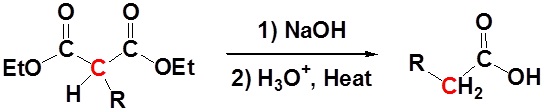
Mechanism
1) Saponification
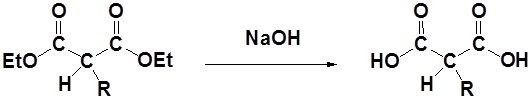
2) Decarboxylation
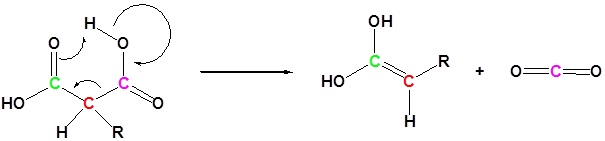
3) Tautomerization

All of the steps together form the Malonic ester synthesis.
\[RX \rightarrow RCH_2CO_2H\]
Example
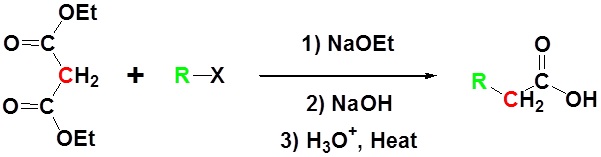
- Bernard E. Hoogenboom , Phillip J. Ihrig , Arne N. Langsjoen , Carol J. Linn and Stephen D. Mulder, The malonic ester synthesis in the undergraduate laboratory, J. Chem. Educ., 1991, 68 (8), p 689
The acetoacetic ester synthesis allows for the conversion of ethyl acetoacetate into a methyl ketone with one or two alkyl groups on the alpha carbon.

Steps
1) Deprotonation with ethoxide

2) Alkylation via and SN2 Reaction

3) Hydrolysis and decarboxylation

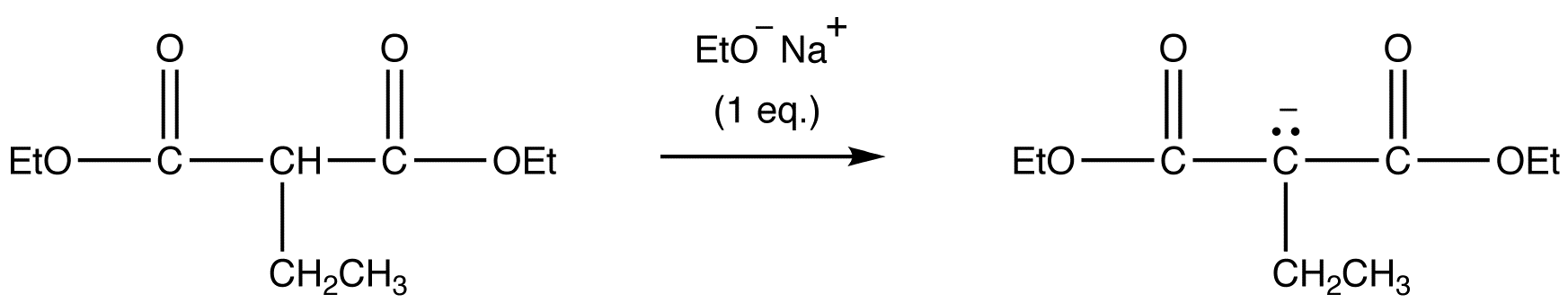
Addition of a second alky group
After the first step and additional alkyl group can be added prior to the decarboxylation step. Overall this allows for the addition of two different alkyl groups.