3.15: Exothermic and Endothermic Processes
- Page ID
- 209986
We first introduced the concept of energy in Module 1 section 8. Recall that energy is manifested in different forms and that it can be converted from one form to another and it can be transferred but must be conserved. In this section we begin to study the energy involved in chemical processes.
A campfire is an example of a combustion reaction and serves as an introduction to thermochemistry. Thermochemistry involves the study of energy changes during chemical reactions. The reaction is initiated by the application of heat from a match. The reaction converting wood to carbon dioxide and water (among other things) continues, releasing heat energy in the process. This heat energy can then be used to cook food, boil water, or just keep warm when it's cold outside.
When physical or chemical changes occur, they are generally accompanied by a transfer of energy. The law of conservation of energy states that in any physical or chemical process, energy is neither created nor destroyed. In other words, the entire energy in the universe is conserved. In order to better understand the energy changes taking place during a reaction, we need to define two parts of the universe, called the system and the surroundings. The system is the specific portion of matter in a given space that is being studied during an experiment or an observation. The surroundings is everything in the universe that is not part of the system. In practical terms for a laboratory chemist, the system is the particular chemicals being reacted, while the surroundings is the immediate vicinity within the room. During most processes, energy is exchanged between the system and the surroundings. If the system loses a certain amount of energy, that same amount of energy is gained by the surroundings. If the system gains a certain amount of energy, that energy is supplied by the surroundings.
A chemical reaction or physical change is endothermic if heat is absorbed by the system from the surroundings. In the course of an endothermic process, the system gains heat from the surroundings and so the temperature of the surroundings decreases. The quantity of heat for a process is represented by the letter \(q\). The sign of \(q\) for an endothermic process is positive because the system is gaining heat. A chemical reaction or physical change is exothermic if heat is released by the system into the surroundings. Because the surroundings is gaining heat from the system, the temperature of the surroundings increases. The sign of \(q\) for an exothermic process is negative because the system is losing heat.
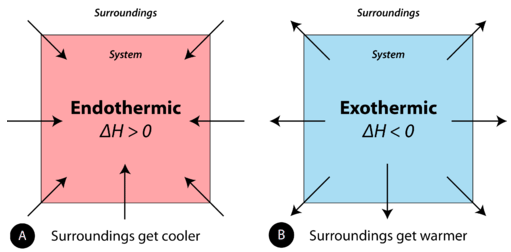
The symbol ΔH, referred to the enthalpy of the reaction, is used in chemistry to represent energy changes under constant pressure conditions (a common condition for reactions that occur in a lab). Read more about enthalpy of reactions in section 3.15.
Energy Diagrams
Energy changes are frequently shown by drawing an energy diagram. Energy diagrams show the potential energy of the reactants and products. If, on an energy diagram, the products have more potential energy than the reactants started with, the reaction is endothermic, energy is added to the reaction. If, on the energy diagram, the products have less potential energy than the reactants started with, the reaction is exothermic, energy is released during the reaction. The “hill” between the reactants and products is the activation energy. This is also referred to as the activation energy barrier because the reactants must have enough energy when they collide to overcome this energy barrier and form products.
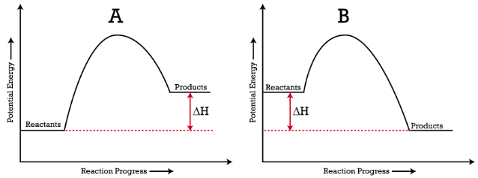
Summary
Chemical reactions involve changes in energy. This may be a change in heat, electricity, light, or other forms of energy. Reactions that absorb energy are endothermic. Reactions that release energy are exothermic.
Contributions & Attributions
This page was constructed from content via the following contributor(s) and edited (topically or extensively) by the LibreTexts development team to meet platform style, presentation, and quality:
Henry Agnew (UC Davis)

