3.17: Temperature and Heat
- Page ID
- 209987
Learning Objectives
- Identify the different between temperature and heat.
- Recognize the different scales used to measure temperature
The concept of temperature may seem familiar to you, but many people confuse temperature with heat. Temperature is a measurement of how hot or cold an object is relative to another object (its thermal energy content). We can measure temperature in the laboratory. Heat is the flow of thermal energy between objects with different temperatures. Temperature is a measure of the average kinetic energy of the particles in matter. In everyday usage, temperature indicates a measure of how hot or cold an object is. Temperature is an important parameter in chemistry. When a substance changes from solid to liquid, it is because there was in increase in the temperature of the material. Chemical reactions usually proceed faster if the temperature is increased. Many unstable materials (such as enzymes) will be viable longer at lower temperatures.

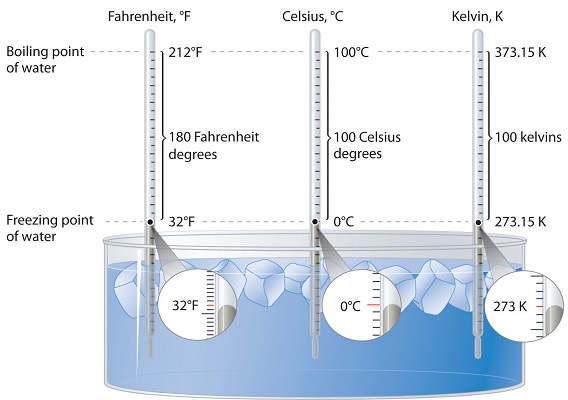
Three different scales are commonly used to measure temperature: Fahrenheit (expressed as °F), Celsius (°C), and Kelvin (K). Thermometers measure temperature by using materials that expand or contract when heated or cooled. Mercury or alcohol thermometers, for example, have a reservoir of liquid that expands when heated and contracts when cooled, so the liquid column lengthens or shortens as the temperature of the liquid changes.
Summary
Three different scales are commonly used to measure temperature: Fahrenheit (expressed as °F), Celsius (°C), and Kelvin (K).
Contributions & Attributions
This page was constructed from content via the following contributor(s) and edited (topically or extensively) by the LibreTexts development team to meet platform style, presentation, and quality:
Henry Agnew (UC Davis)

