9.1 An Introdcution to Acids and Bases
- Page ID
- 218430
Why Do We Care about Acids and Bases?
Heartburn is caused by a buildup of excessive amounts of stomach acid, particularly HCl. This acid is used to digest the food we eat, but it can often back up into the esophagus causing that burning sensation many of us are familiar with. The symptoms of heartburn can be treated with a mild base, which acts to neutralize the excess HCl. For example, Tums is a very commonly used antacid that can be bought over the counter. Other antacids, such as Alka Seltzer, work in similar ways. Below is shown the reaction in which calcium carbonate (the active ingredient in Tums) neutralizes HCl in a simple proton transfer reaction. Following this acid-base reaction, carbonic acid quickly degrades into CO2 and H2O.
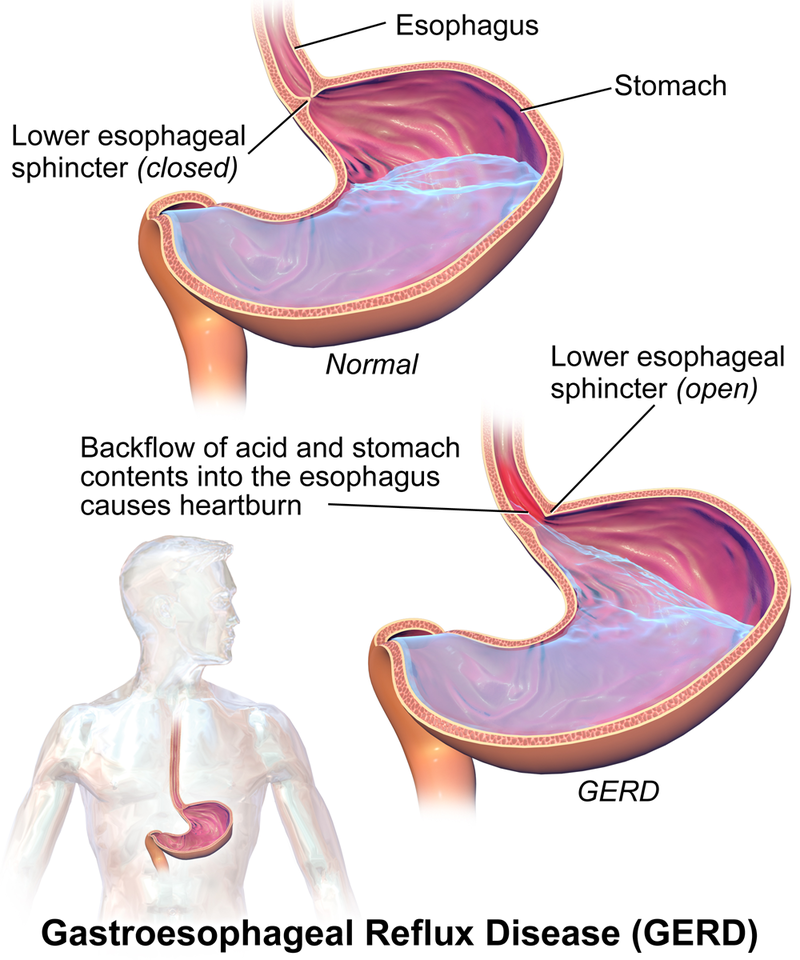
A comparison of a healthy condition to GERD. Image used with permission (Cc BY-SA 4.0; BruceBlaus .
\(CaCO_{3(s)} \; + \; 2HCl_{(aq)} → H_2CO_{3(aq)} \; + \; CaCl_{2(aq)} \)
\(H_2CO_{3(aq)} → H_2O_{(aq)} \; + \; CO_{2(g)} \)
General Descriptions of Acids and Bases
Acids and bases are common in many solutions that exist everywhere, and can be defined by their physical and chemical observations (Table \(\PageIndex{1}\)).
| ACIDS | BASES |
|---|---|
| produce a piercing pain in a wound. | give a slippery feel. |
| taste sour. | taste bitter. |
| are colorless when placed in phenolphthalein (an indicator). | are pink when placed in phenolphthalein (an indicator). |
| are red on blue litmus paper (a pH indicator). | are blue on red litmus paper (a pH indicator). |
| have a pH<7 at 25oC. | have a pH>7 at 25oC. |
| produce carbon dioxide when reacted with carbonates. | |
| Common examples: Lemons, oranges, vinegar, urine, sulfuric acid, hydrochloric acid |
Common Examples: Soap, toothpaste, bleach, cleaning agents, limewater, ammonia water, sodium hydroxide. |
Acids and bases in aqueous solutions will conduct electricity because they contain dissolved ions. Therefore, acids and bases are electrolytes. Strong acids and bases will be strong electrolytes. Weak acids and bases will be weak electrolytes.
The Arrhenius Definition of Acids and Bases
In 1884, the Swedish chemist Svante Arrhenius proposed two specific classifications of compounds, termed acids and bases. When dissolved in an aqueous solution, certain ions were released into the solution. The Arrhenius definition of acid-base reactions is a development of the "hydrogen theory of acids". It was used to provide a modern definition of acids and bases, and followed from Arrhenius's work with Friedrich Wilhelm Ostwald in establishing the presence of ions in aqueous solution in 1884. This led to Arrhenius receiving the Nobel Prize in Chemistry in 1903.
An Arrhenius acid is a compound that increases the concentration of \(H^+\) ions that are present when added to water. These \(H^+\) ions form the hydronium ion (\(H_3O^+\)) when they combine with water molecules. This process is represented in a chemical equation by adding H2O to the reactants side.
\[ HCl_{(aq)} \rightarrow H^+_{(aq)} + Cl^-_{(aq)} \]
In this reaction, hydrochloric acid (\(HCl\)) dissociates into hydrogen (\(H^+\)) and chlorine (\(Cl^-\)) ions when dissolved in water, thereby releasing H+ ions into solution. Formation of the hydronium ion equation:
\[ HCl_{(aq)} + H_2O_{(l)} \rightarrow H_3O^+_{(aq)} + Cl^-_{(aq)} \]
The Arrhenius definitions of acidity and alkalinity are restricted to aqueous solutions and refer to the concentration of the solvated ions. Under this definition, pure \(H_2SO_4\) or \(HCl\) dissolved in acetone are not acidic, despite the fact that both of these acids will donate a proton to acetone. In addition, under the Arrhenius definition, a solution of sodium amide (\(NaNH_2\)) in liquid ammonia is not alkaline, despite the fact that the amide ion (\(NH^−_2\)) will readily deprotonate ammonia. Thus, the Arrhenius definition can only describe acids and bases in an aqueous environment.
Limitation of the Arrhenius Definition of Acids and Bases
The Arrhenius definition can only describe acids and bases in an aqueous environment.

