10.2 Batteries and Electrolytic Cells
- Page ID
- 218435
Batteries
In a battery (also known as a galvanic cell), current is produced when electrons flow externally through the circuit from one substance to the another substance because of a difference in potential energy between the two substances in the electrochemical cell. In a battery made of Zn and Cu, the valence electrons in zinc have a substantially higher potential energy than the valence electrons in copper. Thus, electrons flow spontaneously from zinc to copper(II) ions, forming zinc(II) ions and metallic copper. Just like water flowing spontaneously downhill, which can be made to do work by forcing a waterwheel, the flow of electrons from a higher potential energy to a lower one can also be harnessed to perform work.
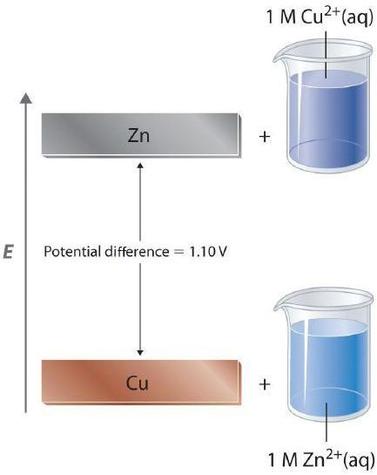
Figure \(\PageIndex{1}\): Potential Energy Difference in the Zn/Cu System. The potential energy of a system consisting of metallic \(Zn\) and aqueous Cu2+ ions is greater than the potential energy of a system consisting of metallic Cu and aqueous \(Zn^{2+}\) ions. Much of this potential energy difference is because the valence electrons of metallic Zn are higher in energy than the valence electrons of metallic Cu. Because the Zn(s) + Cu2+(aq) system is higher in energy by 1.10 V than the Cu(s) + Zn2+(aq) system, energy is released when electrons are transferred from Zn to Cu2+ to form Cu and Zn2+.
Because the potential energy of valence electrons differs greatly from one substance to another, the voltage of a battery depends partly on the identity of the reacting substances. If we construct a battery similar to the one in part (a) in Figure \(\PageIndex{1}\) but instead of copper use a strip of cobalt metal and 1 M Co2+, the measured voltage is not 1.10 V but 0.51 V. Thus we can conclude that the difference in potential energy between the valence electrons of cobalt and zinc is less than the difference between the valence electrons of copper and zinc by 0.59 V.
The measured potential of a cell also depends strongly on the concentrations of the reacting species and the temperature of the system.
A typical battery contains two solid electrodes, which act as the interfaces between a chemical reaction and the external wires through which electrons will flow. There must always be two electrodes because the electrons must be able to travel over a complete circuit. The electrons leave the chemical reaction at the anode, which is the electrode at which oxidation (the loss of electrons) occurs. The electrons travel from the anode, through wires into the device (light bulb, phone, etc.), and then back through more wires to the cathode, which is the electrode at which reduction (the gain of electrons) occurs. Figure \(\PageIndex{2}\) shows a simple battery made from the zinc and copper system described above.
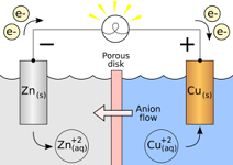
Notice that the electrons carry negative charge through the external wires, but there are no electrons in the battery solution. Inside the battery, ions carry the charge. Anions flow toward the zinc electrode, the electrode at which oxidation occurs. This electrode is called the anode. At the anode, the zinc atoms lose electrons, which leave the battery through the wires. The zinc ions that form enter the solution. The copper cations on the other side of the battery flow towards the other electrode, called the cathode. At the cathode, the copper cations pick up electrons and are reduced to copper metal. The two sides of the battery are kept separate by the porous disk. Only the anions can make it through the disk. If this battery were allowed to run for a long time, the solid zinc anode would become much smaller as zinc atoms were oxidized. At the same time, the copper cathode would get much bigger because copper ions would be getting reduced to form more copper solid on the surface of the cathode.
The lead–acid battery is a common battery used to provide the starting power in virtually every automobile and marine engine on the market. Marine and car batteries typically consist of multiple cells connected in series. The total voltage generated by the battery is the potential per cell (E°cell) times the number of cells.
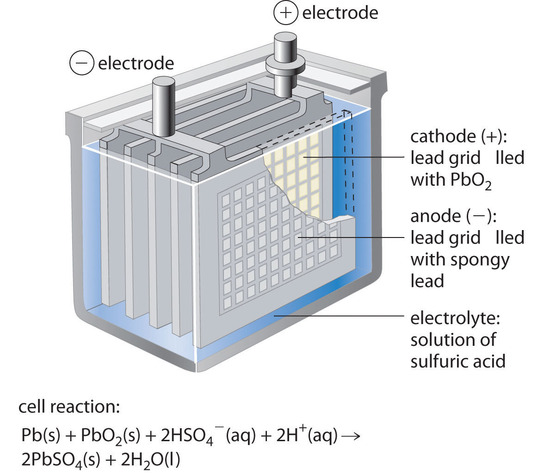
Figure \(\PageIndex{3}\): One Cell of a Lead–Acid Battery. The anodes in each cell of a rechargeable battery are plates or grids of lead containing spongy lead metal, while the cathodes are similar grids containing powdered lead dioxide (PbO2). The electrolyte is an aqueous solution of sulfuric acid. The value of E° for such a cell is about 2 V. Connecting three such cells in series produces a 6 V battery, whereas a typical 12 V car battery contains six cells in series. When treated properly, this type of high-capacity battery can be discharged and recharged many times over.
As shown in Figure \(\PageIndex{3}\), the anode of each cell in a lead storage battery is a plate or grid of spongy lead metal, and the cathode is a similar grid containing powdered lead dioxide (\(PbO_2\)). The electrolyte is usually an approximately 37% solution (by mass) of sulfuric acid in water, with a density of 1.28 g/mL (about 4.5 M \(H_2SO_4\)). Because the redox active species are solids, there is no need to separate the electrodes. The electrode reactions in each cell during discharge are as follows:
- cathode (reduction):
\[PbO_{2(s)} + HSO^−_{4(aq)} + 3H^+_{(aq)} + 2e^− \rightarrow PbSO_{4(s)} + 2H_2O_{(l)} \label{Eq17}\]
with \(E^°_{cathode} = 1.685 \; V\)
- anode (oxidation):
with \(E^°_{anode} = −0.356 \; V\)
-
overall:
\[Pb_{(s)} + PbO_{2(s)} + 2HSO^−_{4(aq)} + 2H^+_{(aq)} \rightarrow 2PbSO_{4(s)} + 2H_2O_{(l)} \label{Eq19}\]
and \(E^°_{cell} = 2.041 \; V\)
Electrolytic Cells
In an electrolytic cell, however, the opposite process, called electrolysis, occurs: an external voltage is applied to drive a nonspontaneous reaction. In this section, we look at how electrolytic cells are constructed and explore one of their many commercial applications.
If we construct an electrochemical cell in which one electrode is copper metal immersed in a 1 M Cu2+ solution and the other electrode is cadmium metal immersed in a \(\,1\; M\, Cd^{2+}\) solution and then close the circuit, the potential difference between the two compartments will be 0.74 V. The cadmium electrode will begin to dissolve (Cd is oxidized to Cd2+) and is the anode, while metallic copper will be deposited on the copper electrode (Cu2+ is reduced to Cu), which is the cathode (Figure \(\PageIndex{1a}\)).
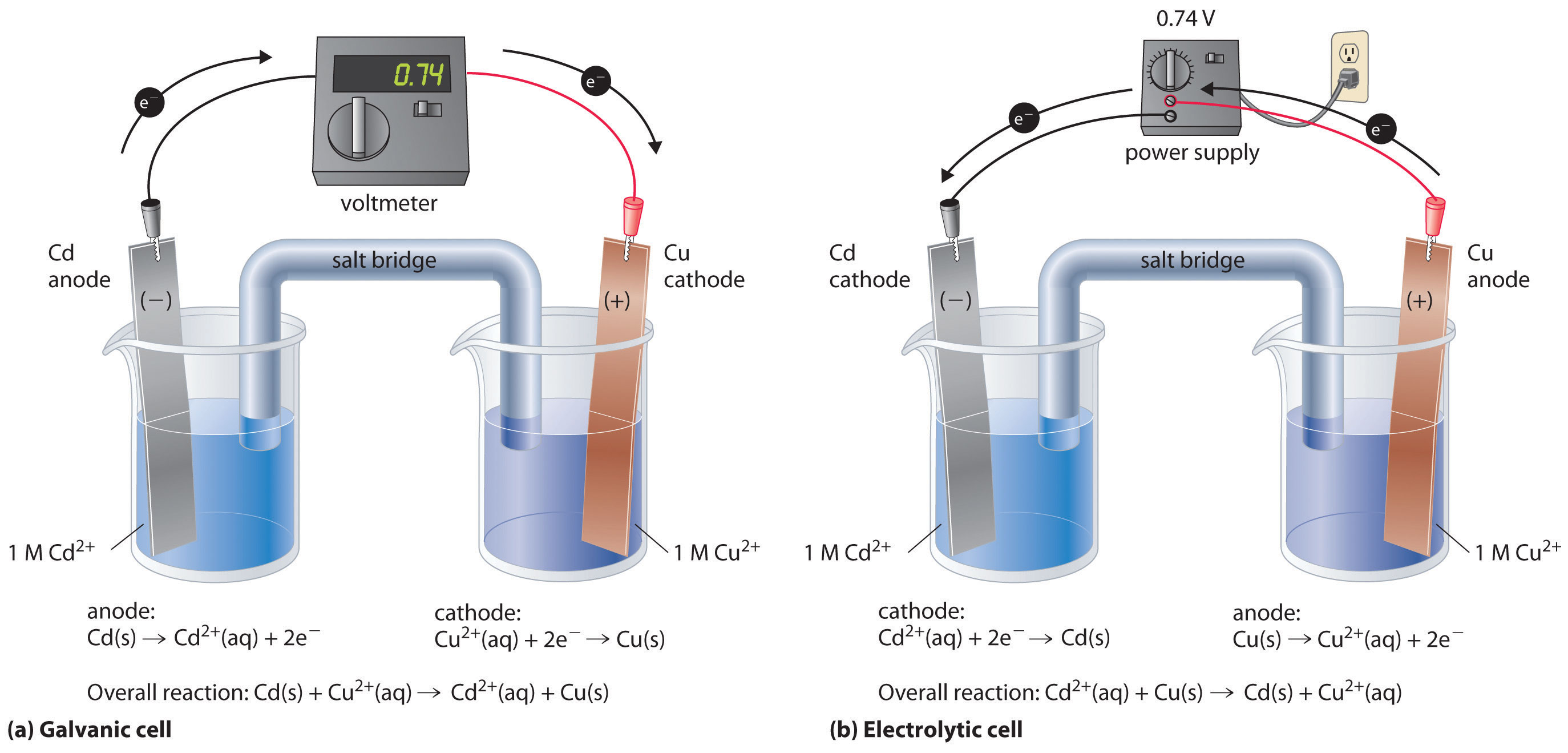
Figure \(\PageIndex{1}\): An Applied Voltage Can Reverse the Flow of Electrons in a Galvanic Cd/Cu Cell. (a) When compartments that contain a Cd electrode immersed in 1 M Cd2+(aq) and a Cu electrode immersed in 1 M Cu2+(aq) are connected to create a galvanic cell, Cd(s) is spontaneously oxidized to Cd2+(aq) at the anode, and Cu2+(aq) is spontaneously reduced to Cu(s) at the cathode. The potential of the galvanic cell is 0.74 V. (b) Applying an external potential greater than 0.74 V in the reverse direction forces electrons to flow from the Cu electrode [which is now the anode, at which metallic Cu(s) is oxidized to Cu2+(aq)] and into the Cd electrode [which is now the cathode, at which Cd2+(aq) is reduced to Cd(s)]. The anode in an electrolytic cell is positive because electrons are flowing from it, whereas the cathode is negative because electrons are flowing into it.
The overall reaction is as follows:
\[ \ce{Cd (s) + Cu^{2+} (aq) \rightarrow Cd^{2+} (aq) + Cu (s)} \label{20.9.1}\]
with \(E°_{cell} = 0.74\; V\)
This reaction is thermodynamically spontaneous as written (ΔG° < 0):
\[ \begin{align} \Delta G^\circ &=-nFE^\circ_\textrm{cell} \label{20.9.2a} \\[5pt] &=-(\textrm{2 mol e}^-)[\mathrm{96,486\;J/(V\cdot mol)}](\mathrm{0.74\;V}) \\[5pt] &=-\textrm{140 kJ (per mole Cd)} \label{20.9.2b} \end{align}\]
In this direction, the system is acting as a galvanic cell.
In an electrolytic cell, an external voltage is applied to drive a nonspontaneous reaction.
The reverse reaction, the reduction of Cd2+ by Cu, is thermodynamically nonspontaneous and will occur only with an input of 140 kJ. We can force the reaction to proceed in the reverse direction by applying an electrical potential greater than 0.74 V from an external power supply. The applied voltage forces electrons through the circuit in the reverse direction, converting a galvanic cell to an electrolytic cell. Thus the copper electrode is now the anode (Cu is oxidized), and the cadmium electrode is now the cathode (Cd2+ is reduced) (Figure \(\PageIndex{1b}\)). The signs of the cathode and the anode have switched to reflect the flow of electrons in the circuit. The half-reactions that occur at the cathode and the anode are as follows:
- cathode:
\[\ce{Cd^{2+}(aq) + 2e^{−} \rightarrow Cd(s)}\label{20.9.3}\]
- anode:
\[\ce{Cu(s) \rightarrow Cu^{2+}(aq) + 2e^{−}} \label{20.9.4}\]
with \(E^°_{anode} = 0.34 \, V \)
- Overall:
\[\ce{Cd^{2+}(aq) + Cu(s) \rightarrow Cd(s) + Cu^{2+}(aq) } \label{20.9.5}\]
with \(E^°_{cell} = −0.74 \: V\)
Because \( E^°_{cell} < 0\), the overall reaction—the reduction of \(Cd^{2+}\) by \(Cu\)—clearly cannot occur spontaneously and proceeds only when sufficient electrical energy is applied.
In a process called electroplating, a layer of a second metal is deposited on the metal electrode that acts as the cathode during electrolysis. Electroplating is used to enhance the appearance of metal objects and protect them from corrosion. Examples of electroplating include the chromium layer found on many bathroom fixtures or (in earlier days) on the bumpers and hubcaps of cars, as well as the thin layer of precious metal that coats silver-plated dinnerware or jewelry. In all cases, the basic concept is the same. A schematic view of an apparatus for electroplating silverware and a photograph of a commercial electroplating cell are shown in Figure \(\PageIndex{4}\).
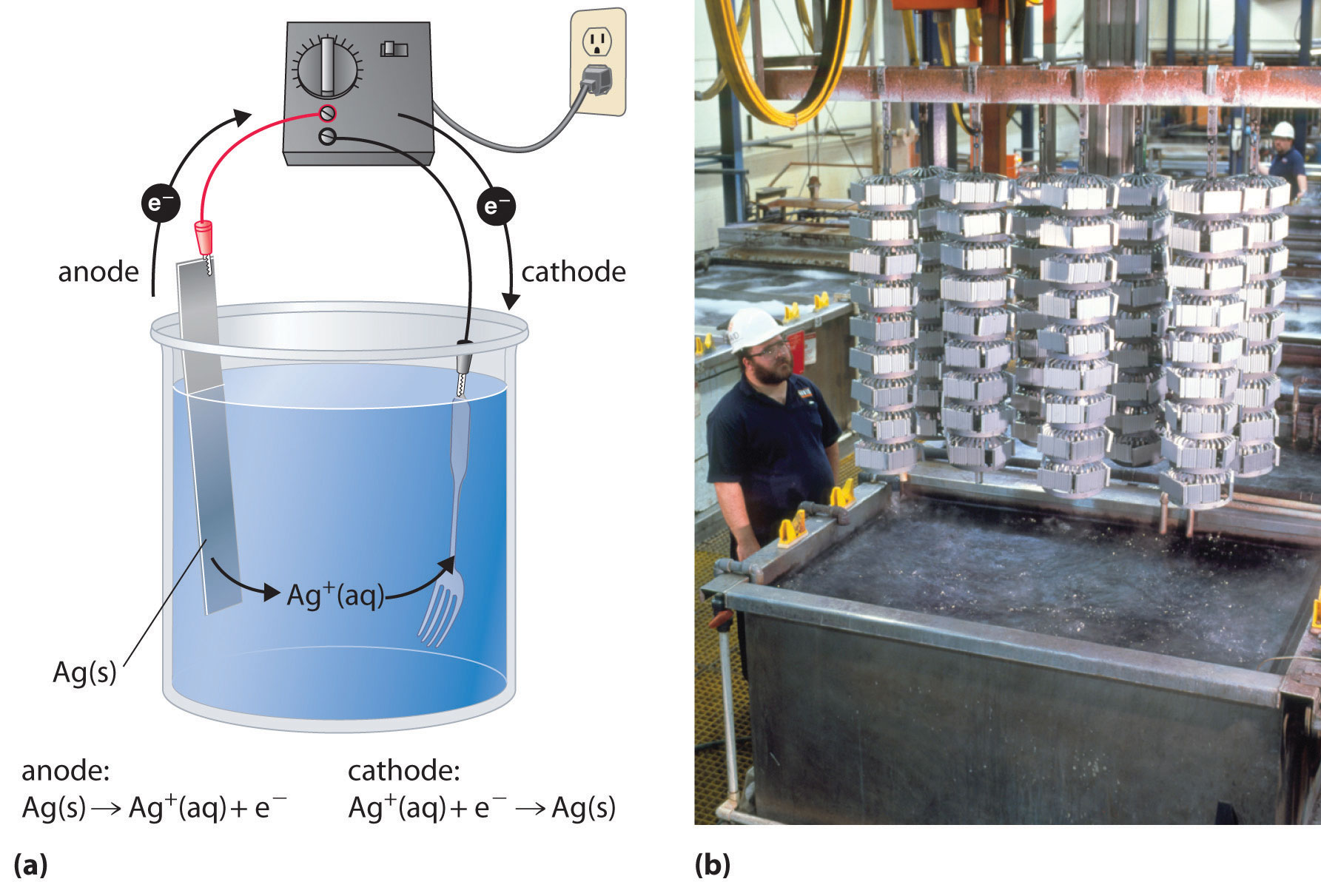
Figure \(\PageIndex{3}\): Electroplating. (a) Electroplating uses an electrolytic cell in which the object to be plated, such as a fork, is immersed in a solution of the metal to be deposited. The object being plated acts as the cathode, on which the desired metal is deposited in a thin layer, while the anode usually consists of the metal that is being deposited (in this case, silver) that maintains the solution concentration as it dissolves. (b) In this commercial electroplating apparatus, a large number of objects can be plated simultaneously by lowering the rack into the Ag+ solution and applying the correct potential.
The half-reactions in electroplating a fork, for example, with silver are as follows:
- cathode (fork): \[Ag^+_{(aq)} + e− \rightarrow Ag_{(s)}\;\;\; E°_{cathode} = 0.80 V\label{20.9.12}\]
- anode (silver bar): \[Ag_{(s)} \rightarrow Ag^+_{(aq)}\;\;\; E°_{anode} = 0.80 V \label{20.9.13}\]
The overall reaction is the transfer of silver metal from one electrode (a silver bar acting as the anode) to another (a fork acting as the cathode). Because E°cell = 0 V, it takes only a small applied voltage to drive the electroplating process. In practice, various other substances may be added to the plating solution to control its electrical conductivity and regulate the concentration of free metal ions, thus ensuring a smooth, even coating.

