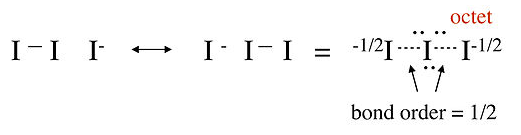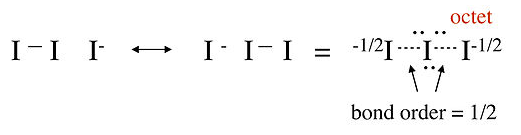The ozone molecule (and related 18e molecules that contain three non-H atoms, such as NO2- and the allyl anion [CH2-CH-CH2]-) is an example of 3-center 4-electron π-bonding. Our MO treatment of ozone is entirely analogous to the 4-electron H3- anion. We map that solution onto this one as follows:
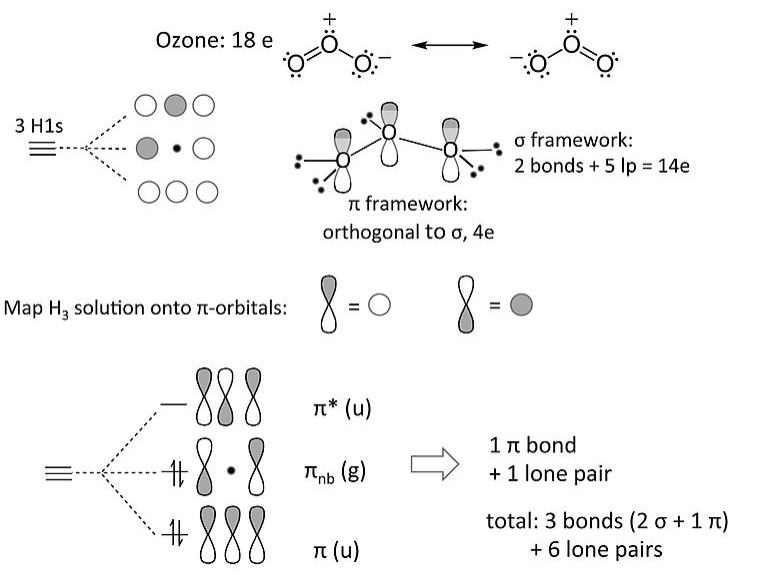
The nonbonding π-orbital has a node at the central O atom. This means that the non-bonding electron pair in the π-system is shared by the two terminal O atoms, i.e., that the formal charge is shared by those atoms. This is consistent with the octet resonance structure of ozone.
This trick of mapping the solution for a set of s-orbitals onto a π-bonding problem is a simple example of a broader principle called the isolobal analogy. This idea, developed extensively by Roald Hoffmann at Cornell University, has been used to understand bonding and reactivity in organometallic compounds.[5] In the isolobal analogy, symmetry principles (as illustrated above in the analogy between H3- and ozone) are used to construct MO diagrams of complex molecules containing d-frontier orbitals from simpler molecular fragments.
|

Professor Roald Hoffmann's ideas about orbital symmetry have helped explain the bonding and reactivity of organic and organometallic molecules, and also the structures and properties of extended solids.
|
The triiodide ion. An analogous (and seemingly more complicated) case of 3-center 4-electron bonding is I3-. Each I atom has 4 valence orbitals (5s, 5px, 5py, 5pz), making a total of 12 frontier orbitals, and the I3- anion has 22 electrons.
We can simplify the problem by recalling two periodic trends:
- The s-p orbital splitting is large, relative to the bond energy, after the second row of the periodic table. Thus, the 5s orbital is low in energy and too contracted to make bonds with its neighbors.
- π-overlap of 5p orbitals is very weak, so the 5px and 5py orbitals will also be non-bonding.
This leaves only the three 5pz orbitals to make bonding/nonbonding/antibonding combinations. Again, the problem is entirely analogous to ozone or H3-.
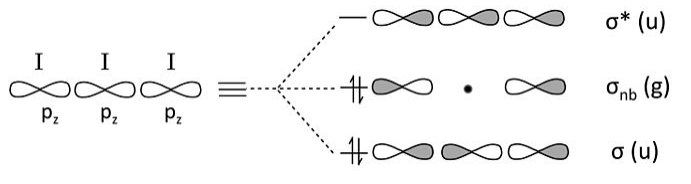
Counting orbitals we obtain 9 lone pairs from the nonbonding 5s, 5px, and 5py orbitals, as well as one bond and one lone pair from the 5pz orbital combinations above. The total of 10 nonbonding pairs and one bond accounts for the 22 electrons in the ion. The non-bonding 5pz pair is localized on the terminal I atoms, giving each a -1/2 formal charge. This MO description is entirely consistent with the octet no-bond resonance picture of I3- that we developed in Chapter 1.