4.3: Reactions of Alkyl Halides- Substitution and Elimination
- Page ID
- 227556
Learning Objective
- apply the alpha and beta labels to alkyl halides for substitution and elimination reactions - refer to section 7.4
Alkyl Halide Structure and Reaction Language
The carbon bonded to a halide is called the alpha-carbon. The carbons bonded to the alpha-carbon are called beta-carbons. Carbon atoms further removed from the alpha carbon are named by continuing the Greek alphabet (alpha, beta, gamma, delta, etc). In discussing the reactions of alkyl halides, it can be effective to use the alpha- and beta- labels. The structure for 2-bromopropane is used below to illustrate the application of these terms.
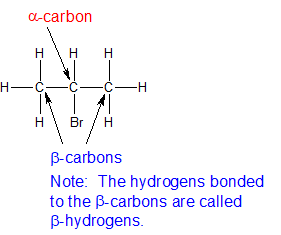
The Reactions - Nucleophilic Substitution and Elimination
Alkyl halides can undergo two major types of reactions - substitution and/or elimination. The substitution reaction is called a Nucleophilic Substitution reaction because the electrophilic alkyl halide forms a new bond with the nucleophile which substitutes for (replaces) the halogen at the alpha-carbon. Because carbon can only form four bonds, the halogen must leave and is called the "Leaving Group". Alkyl halides are excellent electrophiles because halogens share a polar bond with carbon, are polarizable, and form relatively stable leaving groups as halide anions. During a substitution reaction, one sigma bond breaks, and another sigma bond is formed. In the example below, 2-bromopropane is converted into 2-propanol in a substitution reaction.
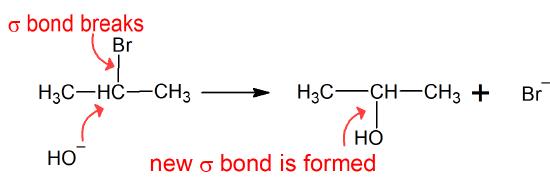
Allkyl halides can also undergo elimination reactions in the presence of strong bases. The elimination of a beta-hydrogen (hydrogen on a carbon vicinal to the alkyl halide carbon) and the halide produces a carbon-carbon double bond to form an alkene. During an elimination reaction, two sigma bonds break, and a pi bond is formed. In the example below, 2-bromopropane has undergone an elimination reaction to give an alkene - propene.
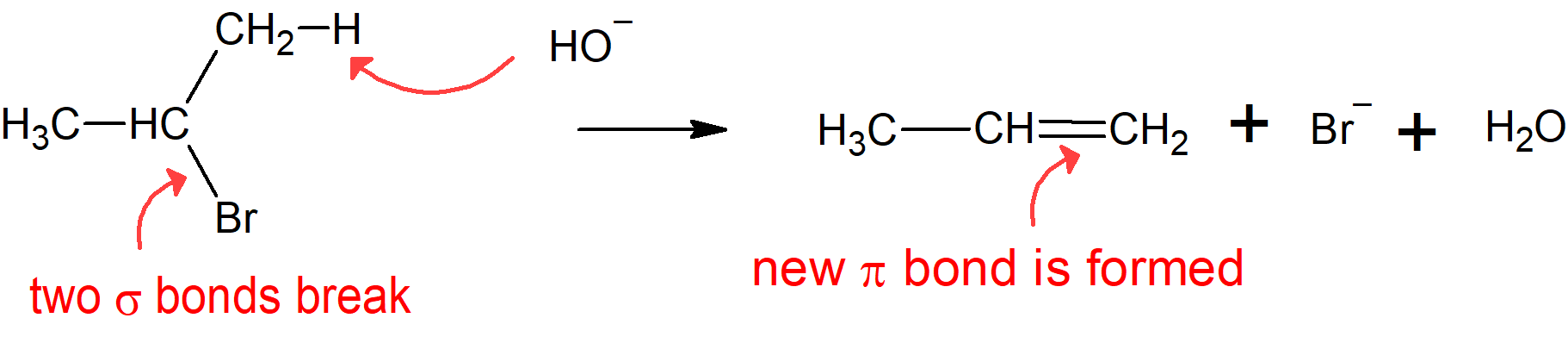
What decides whether you get substitution or elimination?
In the examples above, the reagents were the same for both substitution and elimination - the halogenoalkane and either sodium or potassium hydroxide solution. In all cases, you will get a mixture of both reactions happening - some substitution and some elimination. The product distribution depends on a number of factors. These factors will be explored in the remaining sections of this chapter. Depending on the structure of the alkyl halide, reagent type, reaction conditions, some reactions will only undergo only one pathway - substitution or elimination. While other alkyl halides will always produce a mixture of substitution and elimination products like the example above. The goal of efficient multiple-step synthetic pathways is to maximize the formation of a single product during each step. The reaction conditions explored in this chapter will be useful for future reactions we will study and learn.
Exercise
1. Classify the following reactions as "Substitutions" or "Eliminations".
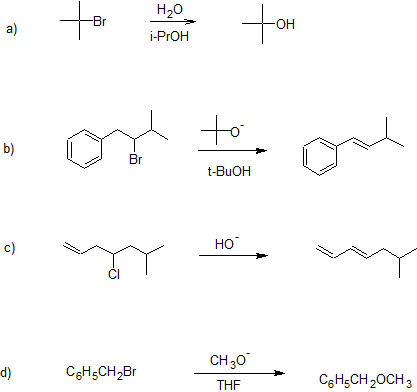
- Answer
-
1. a) substitution
b) elimination
c) elimination
d) substitution
Contributors and Attributions
Dr. Dietmar Kennepohl FCIC (Professor of Chemistry, Athabasca University)
Jim Clark (Chemguide.co.uk)

