2.13: Polyesters
- Page ID
- 272004
Polyester is a category of polymers that contain the ester functional group in every repeat unit of their main chain. The synthesis of polyesters is generally achieved by a polycondensation reaction. The general equation for the reaction of a diol with a diacid is:

General synthesis of a polyester via direct esterification. Image by Minihaa, CC0, via Wikimedia Commons.
Example: poly(ethylene terephthalate) is a polyester obtained between terephthalic acid (benzene-1,4-dicarboxylic acid) and ethylene glycol (ethane-1,2-diol):
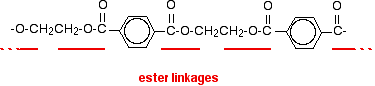 .
.
The everyday name depends on whether it is being used as a fiber or as a material for making things like bottles for soft drinks. When it is being used as a fiber to make clothes, it is often just called polyester. It may sometimes be known by a brand name like Terylene. When it is being used to make bottles, for example, it is usually called PET.
Making polyesters as an example of condensation polymerisation
In condensation polymerisation, when the monomers join together a small molecule gets lost. That's different from addition polymerisation which produces polymers like poly(ethene) - in that case, nothing is lost when the monomers join together. A polyester is made by a reaction involving an acid with two -COOH groups, and an alcohol with two -OH groups. In the common polyester drawn below.
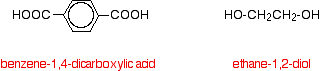
Figure: The acid is benzene-1,4-dicarboxylic acid (old name: terephthalic acid) and the alcohol is ethane-1,2-diol (old name: ethylene glycol).
Now imagine lining these up alternately and making esters with each acid group and each alcohol group, losing a molecule of water every time an ester linkage is made.
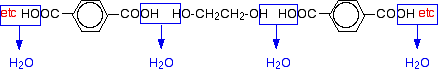
That would produce the chain shown above (although this time written without separating out the carbon-oxygen double bond - write it whichever way you like).
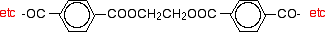
Manufacturing poly(ethylene terephthalate)
The reaction takes place in two main stages: a pre-polymerisation stage and the actual polymerisation. In the first stage, before polymerization happens, you get a fairly simple ester formed between the acid and two molecules of ethane-1,2-diol.
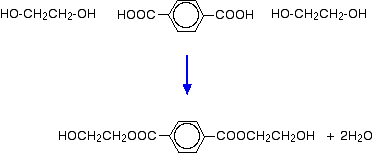
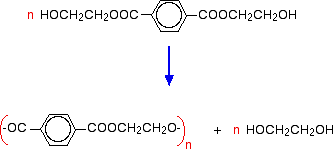
In the polymerisation stage, this is heated to a temperature of about 260°C and at a low pressure. A catalyst is needed - there are several possibilities including antimony compounds like antimony(III) oxide. The polyester forms and half of the ethane-1,2-diol is regenerated. This is removed and recycled.
Hydrolysis of polyesters
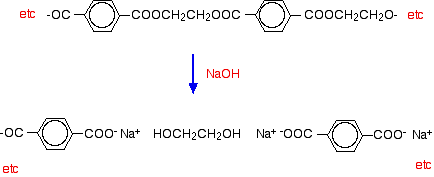
Simple esters are easily hydrolyzed by reaction with dilute acids or alkalis. Polyesters are attacked readily by alkalis, but much more slowly by dilute acids. Hydrolysis by water alone is so slow as to be completely unimportant. (You wouldn't expect your polyester fleece to fall to pieces if you went out in the rain!). If you spill dilute alkali on a fabric made from polyester, the ester linkages are broken. Ethane-1,2-diol is formed together with the salt of the carboxylic acid. Because you produce small molecules rather than the original polymer, the fibers are destroyed, and you end up with a hole! For example, if you react the polyester with sodium hydroxide solution:
Contributors
Jim Clark (Chemguide.co.uk)
Attributions and Citations
- Polyesters. (2020, September 13). Retrieved May 22, 2021, from https://chem.libretexts.org/@go/page/3912
- Wikipedia contributors. (2021, May 20). Polyester. In Wikipedia, The Free Encyclopedia. Retrieved 22:49, May 22, 2021, from https://en.wikipedia.org/w/index.php?title=Polyester&oldid=1024229738


