1.6: Chemical properties II- Reactions of aldehydes and ketones with alcohols
- Page ID
- 227528
In this organic chemistry topic, we shall see how alcohols (R-OH) add to carbonyl groups. Carbonyl groups are characterized by a carbon-oxygen double bond. The two main functional groups that consist of this carbon-oxygen double bond are Aldehydes and Ketones.
Introduction
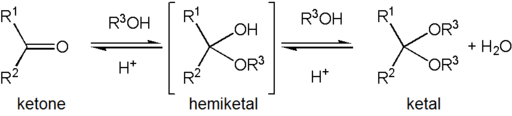
Alcohols add reversibly to aldehydes and ketones to form hemiacetals or hemiketals (hemi, Greek, half). This reaction can continue by adding another alcohol to form an acetal or ketal. These are important functional groups because they appear in sugars.
To achieve effective hemiacetal or acetal formation, two additional features must be implemented. First, an acid catalyst must be used because alcohol is a weak nucleophile; and second, the water produced with the acetal must be removed from the reaction by a process such as a molecular sieves or a Dean-Stark trap. The latter is important, since acetal formation is reversible. Whether the reaction stops at the hemiacetal or hemiketal also depends on the concentration of alcohol used in the experiment. In presence of up to 1 equivalent of alcohol, the reaction stops at the hemiacetal or hemiketal, but in presence of excess of alcohol, the reaction continues to form the acetal and ketal.
Formation of Hemiacetals and Acetals
The term ketal is used to identify the product of the reaction between alcohols and aldehydes (notice that H group from the aldehyde is retained through the reactions)
Figure 1. Formation of a hemiacetal and acetal from the reaction between an aldehyde and an alcohol. Notice that the reaction is reversible and requires an acid catalyst. Image by Ryan Neff, CC BY-SA 3.0, via Wikimedia Commons
Example: reaction between ethanal and ethanol
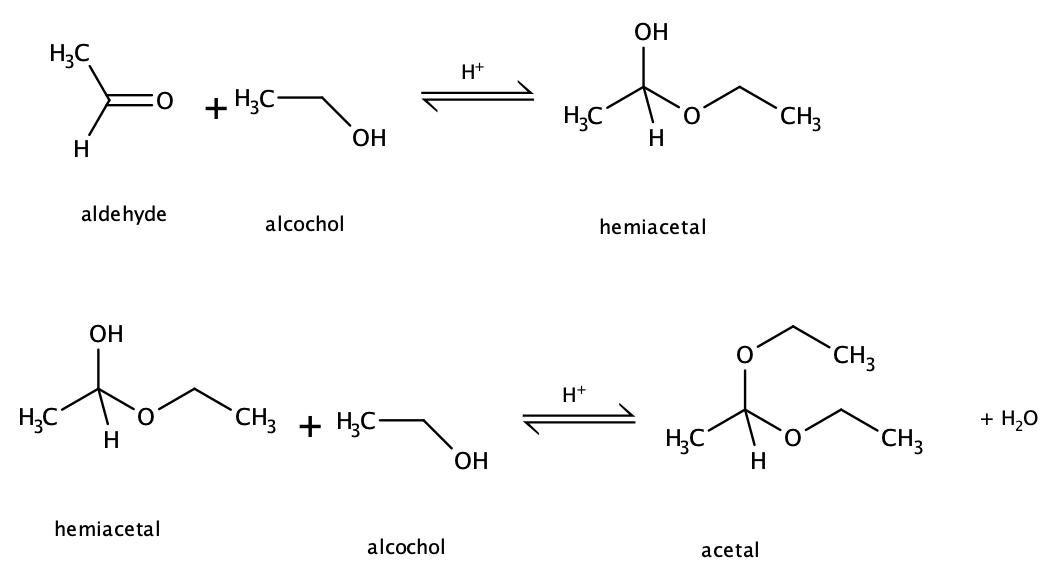
Formation of Hemiketals and ketals
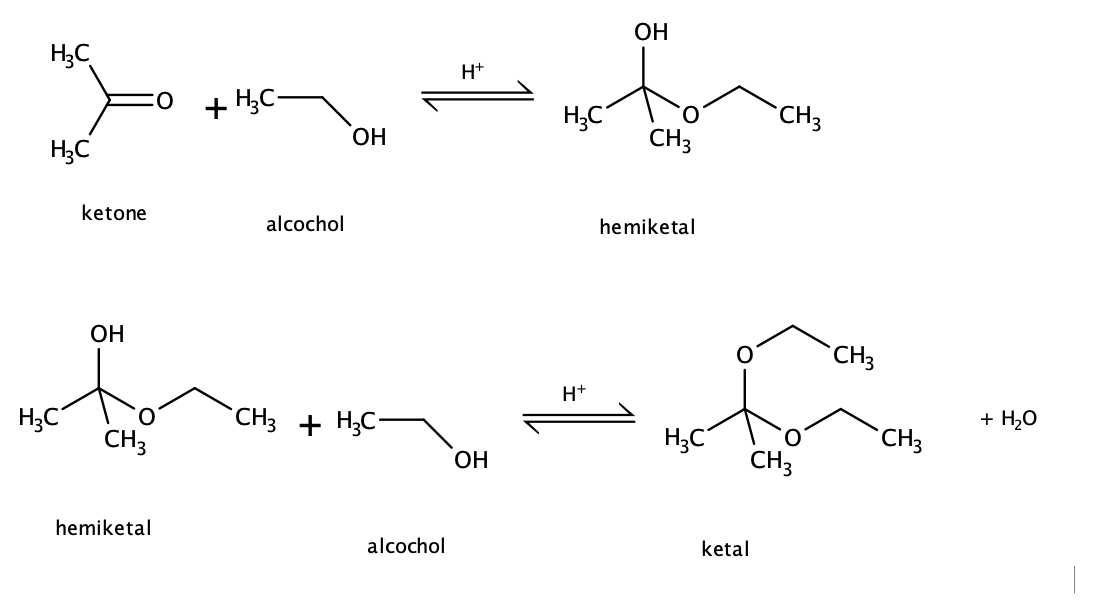
The term ketal is used to identify the product of the reaction between alcohols and ketones (both R groups organic fragments rather than hydrogen)
Figure 2. Formation of a hemiketal and ketal from the reaction between a ketone and an alcohol. Notice that the reaction is reversible and requires an acid catalyst. Image by Ryan Neff, CC BY-SA 3.0, via Wikimedia Commons
Example: reaction between propane and ethanol
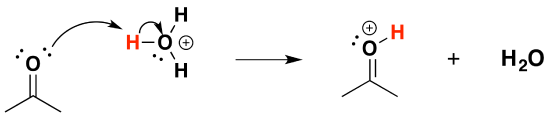
Mechanism for Hemiacetal and Acetal Formation
The mechanism shown here applies to both acetal and hemiacetal formation, but it applies to ketals and hemiketals as well.
a) Formation of an hemiacetal
This reaction is an addition, in which the alcohol molecule behaves as the nucleophile
1) Protonation of the carbonyl
2) Nucleophilic attack by the alcohol
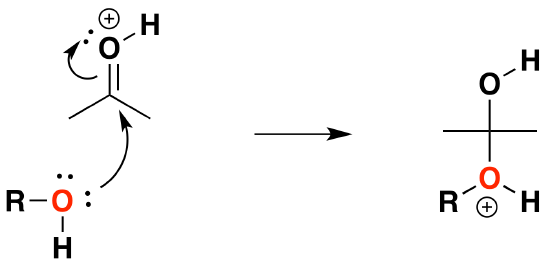
3) Deprotonation to form a hemiacetal
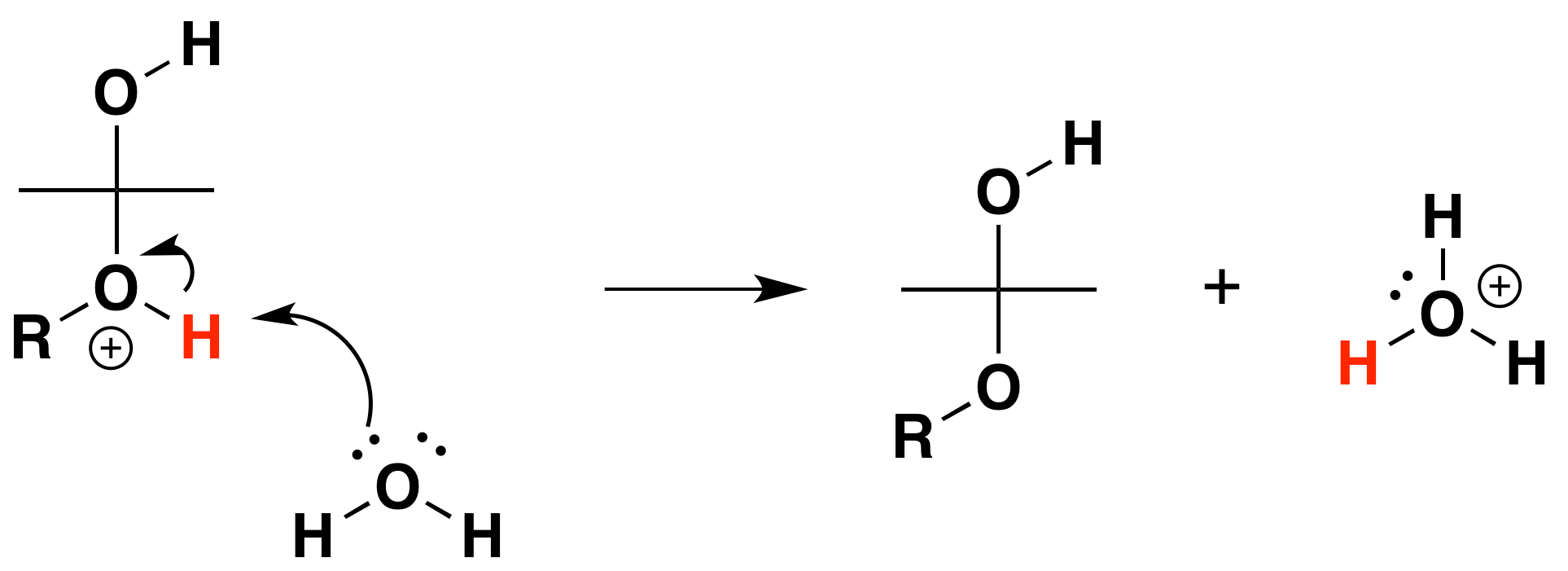
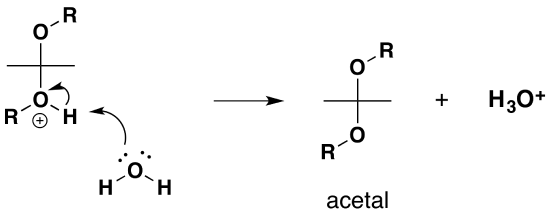
b) Formation of an acetal
This second reaction is a substitution in which the OH group is replace by the RO- from the alcohol
1) Protonation of the alcohol

2) Removal of water
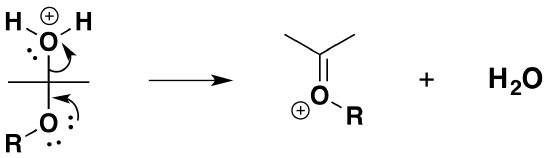
3) Nucleophilic attack by the alcohol
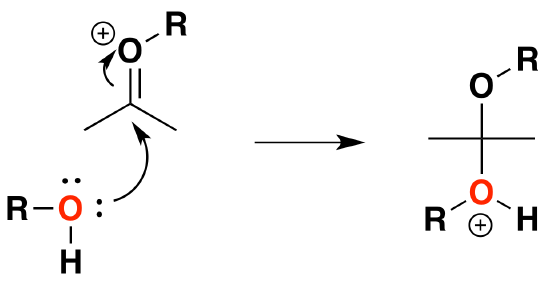
4) Deprotonation by water
Differentiate between acetals, ketals, hemiacetal and hemiketals
The structural similarities between these functional groups might cause some difficulties when identifying whether a given structure corresponds to either one of these functional groups. However, there are some key points to consider that make identification quite easy:
- All four functional groups contain 2 oxygen atoms attached to the same sp3 carbon
- For acetals and hemiacetals, a hydrogen atom remains attached to the sp3 carbon
- For hemiacetals and hemiketals, an OH group remains attached to the sp3 carbon
Example: Identify each product as an acetal, hemiacetal, ketal, or hemiketal:
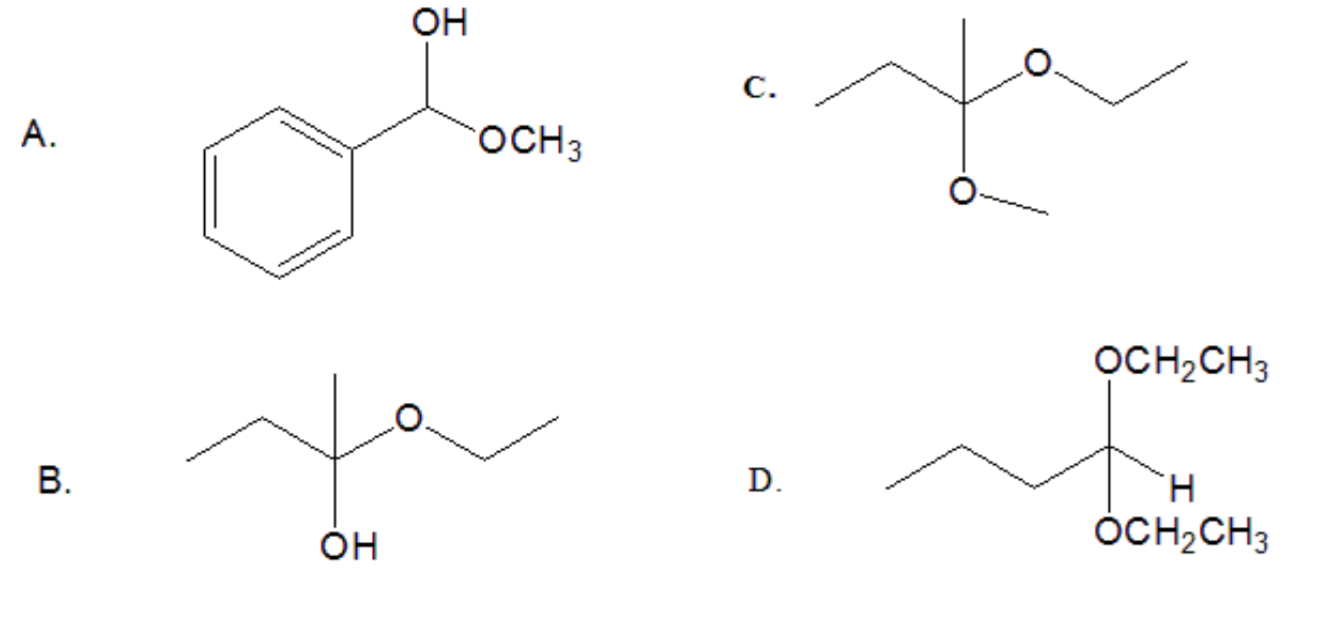
Answer:
a) There is H attached to the sp3 carbon and an OH group. The compound is a hemiacetal
b)There is no H attached to the sp3 carbon and an OH group. The compound is a hemiketal
c) There is no H attached to the sp3 carbon and no OH group. The compound is a ketal
d) There is H attached to the sp3 carbon and no OH group. The compound is a acetal
Formation of Intramolecular (Cyclic) Hemiacetal and Acetals
Molecules which have an alcohol and a carbonyl can undergo an intramolecular reaction to form a cyclic hemiacetal.
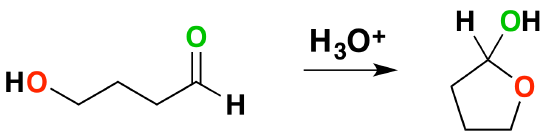
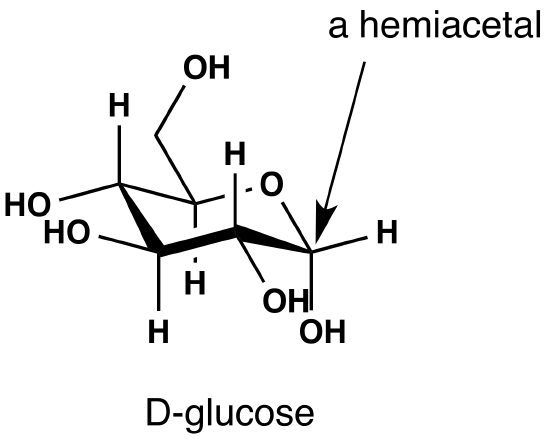
Intramolecular Hemiacetal formation is common in sugar chemistry. For example, the common sugar glucose exists in the cylcic manner more than 99% of the time in a mixture of aqueous solution.
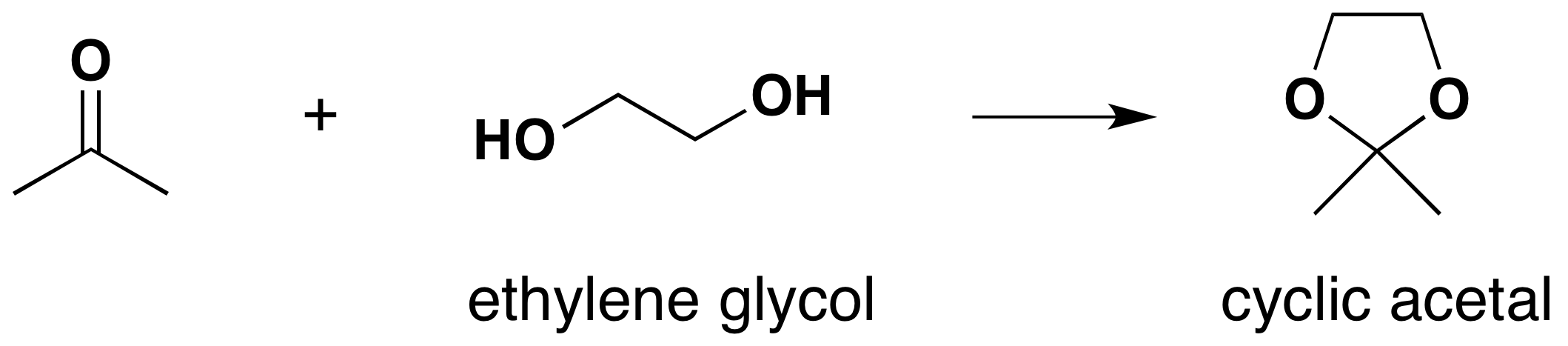
Carbonyls reacting with diol produce a cyclic acetal. A common diol used to form cyclic acetals is ethylene glycol.
Acetals as Protecting Groups
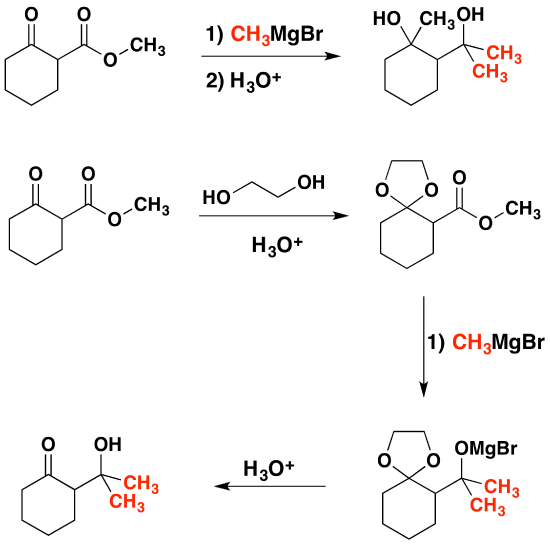
The importance of acetals as carbonyl derivatives lies chiefly in their stability and lack of reactivity in neutral to strongly basic environments. As long as they are not treated by acids, especially aqueous acid, acetals exhibit all the lack of reactivity associated with ethers in general. Among the most useful and characteristic reactions of aldehydes and ketones is their reactivity toward strongly nucleophilic (and basic) metallo-hydride, alkyl and aryl reagents. If the carbonyl functional group is converted to an acetal these powerful reagents have no effect; thus, acetals are excellent protective groups, when these irreversible addition reactions must be prevented.
In the following example we would like a Grignard reagent to react with the ester and not the ketone. This cannot be done without a protecting group because Grignard reagents react with esters and ketones.
References
- Vollhardt, K. Peter C., and Neil E. Schore. Organic Chemistry: Structure and Function. New York: W.H. Freeman and Company, 2007
- Carey, Francis. Advanced Organic Chemistry. 5th ed. Springer, 2007.
- Figure 1 and Figure 2 by Ryan Neff on Wikimedia Commons. Reused under CC BY-SA 3.0 license. No changes were made.
Contributors
Prof. Steven Farmer (Sonoma State University)
William Reusch, Professor Emeritus (Michigan State U.), Virtual Textbook of Organic Chemistry
- Ekram Alexander and Ahmed Rahim (UCD)