10.1: Names and Properties of Alkyl Halides
- Page ID
- 67251
After completing this section, you should be able to
- write the IUPAC name of a halogenated aliphatic hydrocarbon, given its Kekulé, condensed or shorthand structure.
- draw the Kekulé, condensed or shorthand structure of a halogenated aliphatic hydrocarbon, given it IUPAC name.
- write the IUPAC name and draw the Kekulé, condensed or shorthand structure of a simple alkyl halide, given a systematic, non-IUPAC name (e.g., sec-butyl iodide).
- arrange a given series of carbon-halogen bonds in order of increasing or decreasing length and strength.
This section contains little that is new. If you mastered the IUPAC nomenclature of alkanes, you should have little difficulty in naming alkyl halides. Notice that when a group such as CH2Br must be regarded as a substituent, rather than as part of the main chain, we may use terms such as bromomethyl.
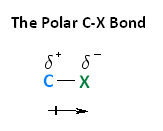
You will find it easier to understand the reactions of the alkyl halides if you keep the polarity of the $\ce{\sf{C-X}}$ bond fixed permanently in your mind (see ”The Polar C$\ce{-}$X Bond” shown in the reading below).
Alkyl halides are also known as haloalkanes. This page explains what they are and discusses their physical properties. alkyl halides are compounds in which one or more hydrogen atoms in an alkane have been replaced by halogen atoms (fluorine, chlorine, bromine or iodine). We will only look at compounds containing one halogen atom. For example:
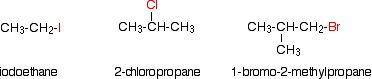
alkyl halides fall into different classes depending on how the halogen atom is positioned on the chain of carbon atoms. There are some chemical differences between the various types.
Primary alkyl halides
In a primary (1°) halogenoalkane, the carbon which carries the halogen atom is only attached to one other alkyl group.Some examples of primary alkyl halides include:
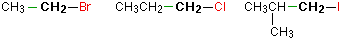
Notice that it doesn't matter how complicated the attached alkyl group is. In each case there is only one linkage to an alkyl group from the CH2 group holding the halogen. There is an exception to this: CH3Br and the other methyl halides are often counted as primary alkyl halides even though there are no alkyl groups attached to the carbon with the halogen on it.
Secondary alkyl halides
In a secondary (2°) halogenoalkane, the carbon with the halogen attached is joined directly to two other alkyl groups, which may be the same or different. Examples:
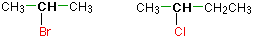
Tertiary alkyl halides
In a tertiary (3°) halogenoalkane, the carbon atom holding the halogen is attached directly to three alkyl groups, which may be any combination of same or different. Examples:
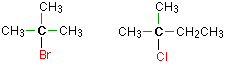
The Learning Objective is to name halogenated hydrocarbons given formulas and write formulas for these compounds given names.
Many organic compounds are closely related to the alkanes. As we noted in Section 12.7, alkanes react with halogens to produce halogenated hydrocarbons, the simplest of which have a single halogen atom substituted for a hydrogen atom of the alkane. Even more closely related are the cycloalkanes, compounds in which the carbon atoms are joined in a ring, or cyclic fashion.
The reactions of alkanes with halogens produce halogenated hydrocarbons, compounds in which one or more hydrogen atoms of a hydrocarbon have been replaced by halogen atoms:

The replacement of only one hydrogen atom gives an alkyl halide (or haloalkane). The common names of alkyl halides consist of two parts: the name of the alkyl group plus the stem of the name of the halogen, with the ending -ide. The IUPAC system uses the name of the parent alkane with a prefix indicating the halogen substituents, preceded by number indicating the substituent’s location. The prefixes are fluoro-, chloro-, bromo-, and iodo-. Thus CH3CH2Cl has the common name ethyl chloride and the IUPAC name chloroethane. Alkyl halides with simple alkyl groups (one to four carbon atoms) are often called by common names. Those with a larger number of carbon atoms are usually given IUPAC names.
Please indicate if the following haloalkanes are methyl, 1o, 2o, or 3o:
a) CH3I
b) CH3CH2Br
c)
d)
e)
f)
Solution
a) methyl
b) 1o
c) 2o
d) 3o
e) 1o
f) 2o
Halogens and the Character of the Carbon-Halogen Bond
With respect to electronegativity, halogens are more electronegative than carbons. This results in a carbon-halogen bond that is polarized. As shown in the image below, carbon atom has a partial positive charge, while the halogen has a partial negative charge.
The following image shows the relationship between the halogens and electronegativity. Notice, as we move up the periodic table from iodine to fluorine, electronegativity increases.
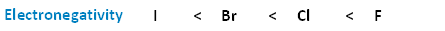
The following image shows the relationships between bond length, bond strength, and molecular size. As we progress down the periodic table from fluorine to iodine, molecular size increases. As a result, we also see an increase in bond length. Conversely, as molecular size increases and we get longer bonds, the strength of those bonds decreases.

Haloalkanes Have Higher Boiling Points than Alkanes
When comparing alkanes and haloalkanes, we will see that haloalkanes have higher boiling points than alkanes containing the same number of carbons. London dispersion forces are the first of two types of forces that contribute to this physical property. You might recall from general chemistry that London dispersion forces increase with molecular surface area. In comparing haloalkanes with alkanes, haloalkanes exhibit an increase in surface area due to the substitution of a halogen for hydrogen. The incease in surface area leads to an increase in London dispersion forces, which then results in a higher boiling point.
Dipole-dipole interaction is the second type of force that contributes to a higher boiling point. As you may recall, this type of interaction is a coulombic attraction between the partial positive and partial negative charges that exist between carbon-halogen bonds on separate haloalkane molecules. Similar to London dispersion forces, dipole-dipole interactions establish a higher boiling point for haloalkanes in comparison to alkanes with the same number of carbons.

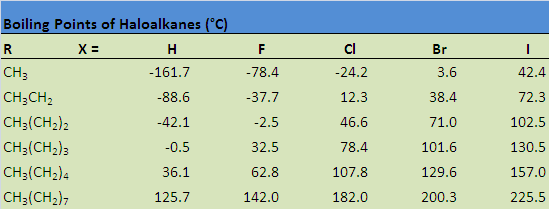
The table below illustrates how boiling points are affected by some of these properties. Notice that the boiling point increases when hydrogen is replaced by a halogen, a consequence of the increase in molecular size, as well as an increase in both London dispersion forces and dipole-dipole attractions. The boiling point also increases as a result of increasing the size of the halogen, as well as increasing the size of the carbon chain.
Solubility
Solubility in water
The alkyl halides are at best only slightly soluble in water. For a halogenoalkane to dissolve in water you have to break attractions between the halogenoalkane molecules (van der Waals dispersion and dipole-dipole interactions) and break the hydrogen bonds between water molecules. Both of these cost energy. Energy is released when ne These will only be dispersion forces and dipole-dipole interactions. These aren't as strong as the original hydrogen bonds in the water, and so not as much energy is released as was used to separate the water molecules. The energetics of the change are sufficiently "unprofitable" that very little dissolves.
Solubility in organic solvents
Alkyl halides tend to dissolve in organic solvents because the new intermolecular attractions have much the same strength as the ones being broken in the separate halogenoalkane and solvent.
Chemical Reactivity
The pattern in strengths o
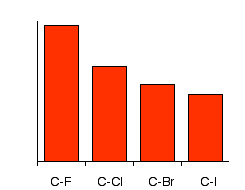
Notice that bond strength orine bond is than the rest. To react with the alkyl halides, the carbon-halogen bond has got to be broken. Because that gets easier as you go from fluoride to chloride to bromide to iodide, the compounds get more reactive in that order. Iodoalkanes are the most reactive and fluoroalkanes are the least. In fact, fluoroalkanes are so unreactive that we shall pretty well ignore them completely from now on in this section!
Exercises
Give the common and IUPAC names for each compound.
- CH3CH2CH2Br
- (CH3)2CHCl
- CH3CH2I
- CH3CH2CH2CH2F
- Answer
-
a. The alkyl group (CH3CH2CH2–) is a propyl group, and the halogen is bromine (Br). The common name is therefore propyl bromide. For the IUPAC name, the prefix for bromine (bromo) is combined with the name for a three-carbon chain (propane), preceded by a number identifying the carbon atom to which the Br atom is attached, so the IUPAC name is 1-bromopropane.
b. The alkyl group [(CH3)2CH–] has three carbon atoms, with a chlorine (Cl) atom attached to the middle carbon atom. The alkyl group is therefore isopropyl, and the common name of the compound is isopropyl chloride. For the IUPAC name, the Cl atom (prefix chloro-) attached to the middle (second) carbon atom of a propane chain results in 2-chloropropane.
c. The alkyl group (CH3CH2–) is a ethyl group, and the halogen is iodide (I). The common name is therefore ethyl iodide. For the IUPAC name, the prefix for Iodide (Iodod) is combined with the name for a two-carbon chain (ethane), preceded by a number identifying the carbon atom to which the I atom is attached, so the IUPAC name is 1-bromoethane
d. The alkyl group (CH3CH2CH2CH2–) is a butyl group, and the halogen is fluorine (F). The common name is therefore butyl fluoride. For the IUPAC name, the prefix for Fluorine (Fluoro) is combined with the name for a four-carbon chain (butane), preceded by a number identifying the carbon atom to which the Br atom is attached, so the IUPAC name is 1-fluorobutane.
Give the IUPAC name for each compound.
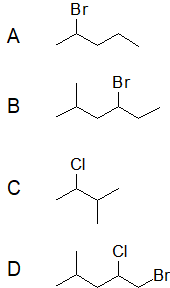
- Answer
-
A) The parent alkane has five carbon atoms in the longest continuous chain; it is pentane. A bromo (Br) group is attached to the second carbon atom of the chain. The IUPAC name is 2-bromopentane.
B) The parent alkane is hexane. Methyl (CH3) and bromo (Br) groups are attached to the second and fourth carbon atoms, respectively. Listing the substituents in alphabetical order gives the name 4-bromo-2-methylhexane.
C) 2-Chloro-3-methylbutane
D) 1-Bromo-2-chloro-4-methylpentane rine (F). The common name is therefore butyl fluoride. For the IUPAC name, the prefix for Fluorine (Fluoro) is combined with the name for a four-carbon chain (butane), preceded by a number identifying the carbon atom to which the Br atom is attached, so the IUPAC name is 1-fluorobutane.
Give the names of the following organohalides:
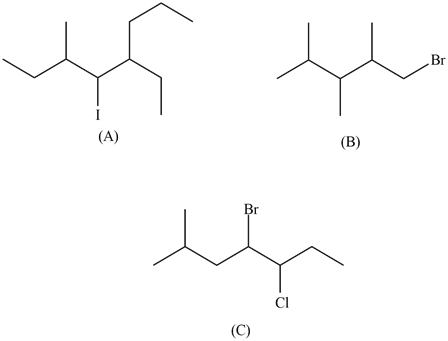
- Answer
-
(A) 5-ethyl-4-iodo-3methyl-octane
(B) 1-bromo-2,3,4-trimethyl-pentane
(C) 4-bromo-5-chloro-2-methyl-heptane
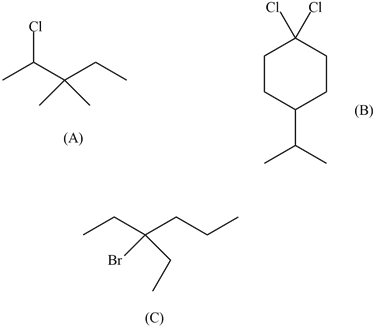
Draw the structures of the following compounds:
(A) 2-Chloro-3,3-dimethylpentane
(B) 1,1-Dichloro-4-isopropylcyclohexane
(C) 3-bromo-3-ethylhexane
- Answer
-
Contributors and Attributions
Dr. Dietmar Kennepohl FCIC (Professor of Chemistry, Athabasca University)
Prof. Steven Farmer (Sonoma State University)
Jim Clark (Chemguide.co.uk)
- Rachael Curtis (UC Davis)

