Bonding in Benzene - a Modern Orbital View
- Page ID
- 3640
Building the orbital model
Benzene is built from hydrogen atoms (1s1) and carbon atoms (1s22s22px12py1). Each carbon atom has to join to three other atoms (one hydrogen and two carbons) and doesn't have enough unpaired electrons to form the required number of bonds, so it needs to promote one of the 2s2 pair into the empty 2pz orbital.
Figure 1.1: Step 1: Promotion of an electron
There is only a small energy gap between the 2s and 2p orbitals, and an electron is promoted from the 2s to the empty 2p to give 4 unpaired electrons. The extra energy released when these electrons are used for bonding more than compensates for the initial input. The carbon atom is now said to be in an excited state.
Because each carbon is only joining to three other atoms, when the carbon atoms hybridize their outer orbitals before forming bonds, they only need to hybridise three of the orbitals rather than all four. They use the 2s electron and two of the 2p electrons, but leave the other 2p electron unchanged.'
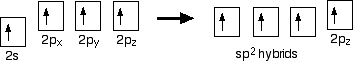
Figure 1.2: Step 2: Hydribidization of the atomic orbitals
The new orbitals formed are called sp2 hybrids, because they are made by an s orbital and two p orbitals reorganizing themselves. The three sp2 hybrid orbitals arrange themselves as far apart as possible - which is at 120° to each other in a plane. The remaining p orbital is at right angles to them. Each carbon atom now looks like the diagram above. This is all exactly the same as happens in ethene.
The difference in benzene is that each carbon atom is joined to two other similar carbon atoms instead of just one. Each carbon atom uses the sp2 hybrids to form sigma bonds with two other carbons and one hydrogen atom. The next diagram shows the sigma bonds formed, but for the moment leaves the p orbitals alone.
Only a part of the ring is shown because the diagram gets extremely cluttered if you try to draw any more. Notice that the p electron on each carbon atom is overlapping with those on both sides of it. This extensive sideways overlap produces a system of pi bonds which are spread out over the whole carbon ring. Because the electrons are no longer held between just two carbon atoms, but are spread over the whole ring, the electrons are said to be delocalized. The six delocalized electrons go into three molecular orbitals - two in each.
In common with the great majority of descriptions of the bonding in benzene, we are only going to show one of these delocalized molecular orbitals for simplicity.
In the diagram, the sigma bonds have been shown as simple lines to make the diagram less confusing. The two rings above and below the plane of the molecule represent one molecular orbital. The two delocalized electrons can be found anywhere within those rings. The other four delocalized electrons live in two similar (but not identical) molecular orbitals.\
The Symbol for benzene
Although you will still come across the Kekulé structure for benzene, for most purposes we use the structure with a hexagon showing the ring of six carbon atoms, each of which has one hydrogen attached. (You have to know that - counting bonds to find out how many hydrogens to add doesn't work in this particular case.)
The circle represents the delocalized electrons. It is essential that you include the circle. If you miss it out, you are drawing cyclohexane and not benzene.
Relating Electronic Structrure to Properties of Benzene
- The shape of benzene: Benzene is a planar regular hexagon, with bond angles of 120°. This is easily explained. It is a regular hexagon because all the bonds are identical. The delocalization of the electrons means that there aren't alternating double and single bonds. It is planar because that is the only way that the p orbitals can overlap sideways to give the delocalized \(\pi\) system.
- The energetic stability of benzene: This is accounted for by the delocalization. As a general principle, the more you can spread electrons around - in other words, the more they are delocalized - the more stable the molecule becomes. The extra stability of benzene is often referred to as "delocalization energy".
- The reluctance of benzene to undergo addition reactions: With the delocalized electrons in place, benzene is about 150 kJ mol-1 more stable than it would otherwise be. If you added other atoms to a benzene ring you would have to use some of the delocalized electrons to join the new atoms to the ring. That would disrupt the delocalization and the system would become less stable. Since about 150 kJ per mole of benzene would have to be supplied to break up the delocalization, this isn't going to be an easy thing to do.
Contributors
Jim Clark (Chemguide.co.uk)


