Carboxylic Derivatives - The Claisen Condensation
- Page ID
- 1220
Claisen Condensation
The aldol reaction, is a remarkable and useful reaction of aldehydes and ketones in which the carbonyl group serves both as an electrophilic reactant and the source of a nucleophilic enol species. Esters undergo a similar transformation called the Claisen Condensation. Four examples of this base-induced reaction, which usually forms beta-ketoester products, are shown in the following diagram. Greek letter assignments for the ester products are given in blue. Equation #1 presents the synthesis of the important reagent ethyl acetoacetate, and #2 illustrates the general form of the Claisen condensation. Intramolecular reactions, such as #3, lead to rings (usually five or six-membered) and are referred to as Dieckmann Condensations. The last equation shows a mixed condensation between two esters, one of which has no alpha-hydrogens. The product in this case is a phenyl substituted malonic ester rather than a ketoester.
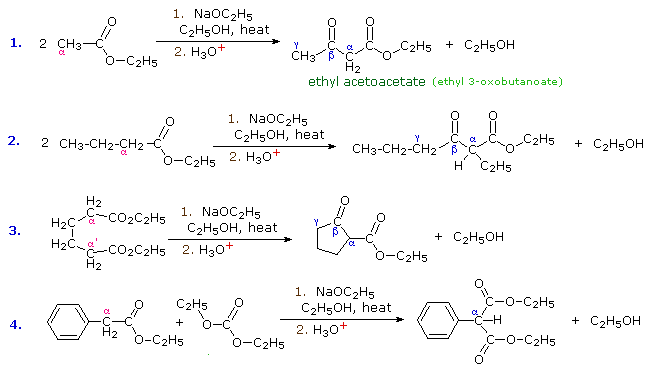 |
|||
By clicking the "Structural Analysis" button below the diagram, a display showing the nucleophilic enolic donor molecule and the electrophilic acceptor molecule together with the newly formed carbon-carbon bond will be displayed. A stepwise mechanism for the reaction will be shown by clicking the "Reaction Mechanism" button. In a similar mode to the aldol reaction, the fundamental event in the Claisen condensation is a dimerization of two esters by an alpha C–H addition of one reactant to the carbonyl group of a second reactant. This bonding is followed by alcohol elimination from the resulting hemiacetal. The eventual formation of a resonance stabilized beta-ketoester enolate anion, as shown on the third row of the mechanism, provides a thermodynamic driving force for the condensation. Note that this stabilization is only possible if the donor has two reactive alpha-hydrogens.
The Claisen condensation differs from the aldol reaction in several important ways.
(i) The aldol reaction may be catalyzed by acid or base, but most Claisen condensations require base.
(ii) In contrast to the catalytic base used for aldol reactions, a full equivalent of base (or more) must be used for the Claisen condensation. The extra base is needed because beta-ketoesters having acidic hydrogens at the alpha-carbon are stronger acids (by about 5 powers of ten) than the alcohol co-product. Consequently, the alkoxide base released after carbon-carbon bond formation (upper right structure in the mechanism diagram) immediately removes an alpha proton from the beta-ketoester product. As noted above, formation of this doubly-stabilized enolate anion provides a thermodynamic driving force for the condensation.
(iii) The aldol reaction may be catalyzed by hydroxide ion, but the Claisen condensation requires that alkoxide bases be used, in order to avoid ester hydrolysis. The specific alkoxide base used should match the alcohol component of the ester to avoid ester exchange reactions. Very strong bases such as LDA may also be used in this reaction.
(iv) The stabilized enolate product must be neutralized by aqueous acid in order to obtain the beta-ketoester product.
Transformations similar to the Claisen condensation may be effected with mixed carbonyl reactants, which may include ketones and nitriles as well as esters. Esters usually serve as the electrophilic acceptor component of the condensation. Acyl chlorides and anhydrides would also be good electrophilic acceptors, but they are more expensive than esters and do not tolerate the alcohol solvents often used for Claisen condensations.
In the case of mixed condensations, complex product mixtures are commonly avoided by using an acceptor ester that has no alpha-hydrogens. Examples of such reactants are: ethyl formate (HCO2C2H5), diethyl carbonate (C2H5OCO2C2H5), ethyl benzoate (C6H5CO2C2H5) and diethyl oxalate (C2H5O2C-CO2C2H5). Equations #2, 3 & 4 below illustrate the use of such acceptors with ester, ketone and nitrile donor compounds. The nucleophilic enol species from the nitrile in #4 may be written as: C6H5CH=C=N(–). The 2-formylcyclohexanone product from reaction #3 exists predominantly as its hydrogen-bonded enol. Most beta-ketoesters have significant enol concentrations, but the formyl group has an exceptional bias for this tautomer.
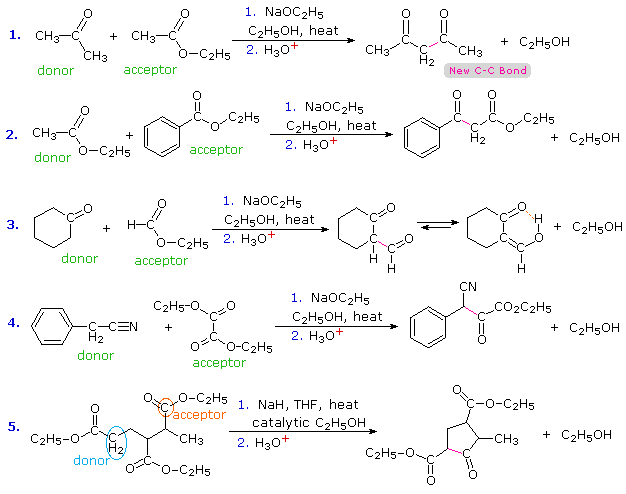
Equation #1 shows a condensation in which both reactants might serve either as donors or acceptors. The selective formation of one of the four possible condensation products is due to the reversibility of these reactions and the driving force provided by resonance stabilization of the enolate anion of 2,4-pentanedione (pKa=9). Protonation of this anion gives the product. The last equation (#5) presents an interesting example of selectivity. There are three ester functions, each of which has at least one alpha-hydrogen. Only one of these, that on the left, has two alpha-hydrogens and will yield an enolizable beta-ketoester by functioning as the donor in a Dieckmann cyclization. Strained four-membered rings are not favored by reversible condensation reactions, so ring closure to the ester drawn below the horizontal chain does not occur. The only reasonable product is the five-membered cyclic ketoester.
Although many Claisen condensations are carried out with a full equivalent of the alkoxide base, an effective alternative procedure, used in reaction #5, uses sodium hydride (NaH) together with a catalytic amount of alcohol. The catalytic alcohol reacts with NaH to produce alkoxide, this initiates a condensation reaction and the product alcohol then reacts with more NaH to give alkoxide.


