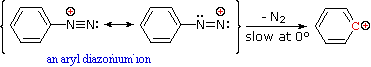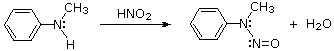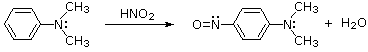Reaction of Amines with Nitrous Acid
- Page ID
- 770
Nitrous acid (\(HNO_2\) or \(HONO\)) reacts with aliphatic amines in a fashion that provides a useful test for distinguishing primary, secondary and tertiary amines.
- 1°-Amines + HONO (cold acidic solution) \(\rightarrow\) Nitrogen Gas Evolution from a Clear Solution
- 2°-Amines + HONO (cold acidic solution) \(\rightarrow\) An Insoluble Oil (N-Nitrosamine)
- 3°-Amines + HONO (cold acidic solution) \(\rightarrow\) A Clear Solution (Ammonium Salt Formation)
Nitrous acid is a Brønsted acid of moderate strength (pKa = 3.3). Because it is unstable, it is prepared immediately before use in the following manner:
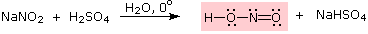
Under the acidic conditions of this reaction, all amines undergo reversible salt formation:

This happens with 3º-amines, and the salts are usually soluble in water. The reactions of nitrous acid with 1°- and 2°- aliphatic amines may be explained by considering their behavior with the nitrosonium cation, NO(+), an electrophilic species present in acidic nitrous acid solutions.
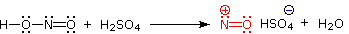
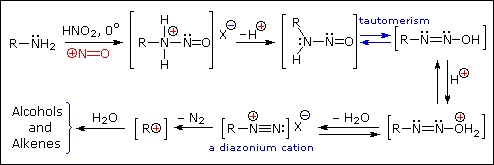
Primary Amines
Secondary Amines
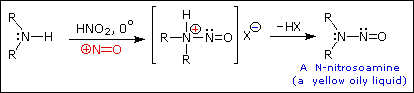
The distinct behavior of 1º, 2º & 3º-aliphatic amines is an instructive challenge to our understanding of their chemistry, but is of little importance as a synthetic tool. The SN1 product mixtures from 1º-amines are difficult to control, and rearrangement is common when branched primary alkyl groups are involved. The N-nitrosamines formed from 2º-amines are carcinogenic, and are not generally useful as intermediates for subsequent reactions.
Aryl Amines
Nitrous acid reactions of 1º-aryl amines generate relatively stable diazonium species that serve as intermediates for a variety of aromatic substitution reactions. Diazonium cations may be described by resonance contributors, as in the bracketed formulas shown below. The left-hand contributor is dominant because it has greater bonding. Loss of nitrogen is slower than in aliphatic 1º-amines because the C-N bond is stronger, and aryl carbocations are comparatively unstable.
Aqueous solutions of these diazonium ions have sufficient stability at 0º to 10 ºC that they may be used as intermediates in a variety of nucleophilic substitution reactions. For example, if water is the only nucleophile available for reaction, phenols are formed in good yield.
2º-Aryl Amines:
2º-Aryl amines give N-nitrosamine derivatives on reaction with nitrous acid, and thus behave identically to their aliphatic counterparts.
3º-Aryl Amines:
Depending on ring substitution, 3º-Aryl amines may undergo aromatic ring nitrosation at sites ortho or para to the amine substituent. The nitrosonium cation is not sufficiently electrophilic to react with benzene itself, or even toluene, but highly activated aromatic rings such as amines and phenols are capable of substitution. Of course, the rate of reaction of NO(+) directly at nitrogen is greater than that of ring substitution, as shown in the previous example. Once nitrosated, the activating character of the amine nitrogen is greatly diminished; and N-nitrosoaniline derivatives, or indeed any amide derivatives, do not undergo ring nitrosation.
Contributors
- William Reusch, Professor Emeritus (Michigan State U.), Virtual Textbook of Organic Chemistry