Properties and Bonding in the Alkynes
- Page ID
- 910
The characteristic of the triple bond helps to explain the properties and bonding in the alkynes.
Importance of Triple Bonds
Hybridization due to triple bonds allows the uniqueness of alkyne structure. This triple bond contributes to the nonpolar bonding strength, linear, and the acidity of alkynes. Physical Properties include nonpolar due to slight solubility in polar solvents and insoluble in water. This solubility in water and polar solvents is a characteristic feature to alkenes as well. Alkynes dissolve in organic solvents.
Boiling Points
Compared to alkanes and alkenes, alkynes have a slightly higher boiling point. Ethane has a boiling point of -88.6 ?C, while Ethene is -103.7 ?C and Ethyne has a higher boiling point of -84.0 ?C.
Acidity
The acidity of terminal alkynes compared to alkenes and alkanes are stronger. Compared with Ethane which has a pKa of 62 (least acidic) and Ethene of a pKa of 45, Ethyne has a pKa of 26. With alkynes having the sp hybridization, this makes it the most acidic hydrocarbon. Therefore terminal alkynes must be deprotonated by stronger bases. The importance of the s orbital being attracted to the nucleus contributes to the electronegativity
Bonding and Hybridization
| Bond | Name | Location | Overlap |
| Bond 1 | s (? bond) bond | Formed between 2 sp orbitals of carbon and hydrogen atoms | End-on overlap |
| Bond 2 | S (? bond) bond | Formed between the 2 sp orbital of 2 unsaturated Carbon atoms. | End-on overlap |
| Bond 3 | p-bonds (? bonds) | Formed between the 2 p-orbitals among the carbon atoms | Side-on overlap |

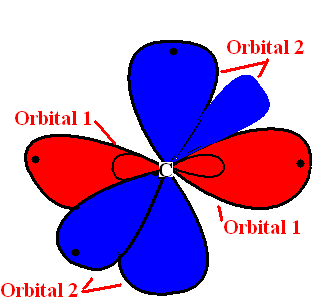
| Orbital | Name |
Location |
| Orbital 1 | sp hybrid orbitals | Formed in the linear structure model of carbon atom |
| Orbital 2 | p-orbitals | Formed on each carbon |
Alkynes are High In Energy
Alkynes are involved in a high release of energy because of repulsion of electrons. The content of energy involved in the alkyne molecule contributes to this high amount of energy. The pi-bonds however, do not encompass a great amount of energy even though the concentration is small within the molecule. The combustion of Ethyne is a major contributor from CO2, water, and the ethyne molecule
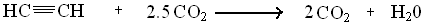
??H = -311 kcal/mol
To help understand the relative stabilities of alkyne isomers, heats of hydrogenation must be used. Hydrogenation of the least energy, results in the release of the internal alkyne. With the result of the production of butane, the stability of internal versus terminal alkynes has significant relative stability due to hyperconjugation.
Outside links
References
- Bloch, D.R. Organic chemistry demystified, New York : McGraw-Hill, 2006.
- Vollhardt. Schore, Organic Chemistry Structure and Function Fifth Edition, New York: W.H. Freeman and Company, 2007.
Problems
- What is the carbon-carbon, carbon-hydrogen bond length for alkyne? Is it shorter or longer than alkane and alkene?
- Which is the most acidic and most stable, alkane, alkene, or alkyne? And depends on what?
- How many pi bonds and sigma bonds are involved in the structure of ethyne?
- Why is the carbon-hydrogen bond so short?
- What is the alkyne triple bond characterizes by? How is this contribute to the weakness of the pi bonds?
- How is heat of hydrogenation effects the stability of the alkyne?
Contributors
- Bao Kha Nguyen, Garrett M. Chin

