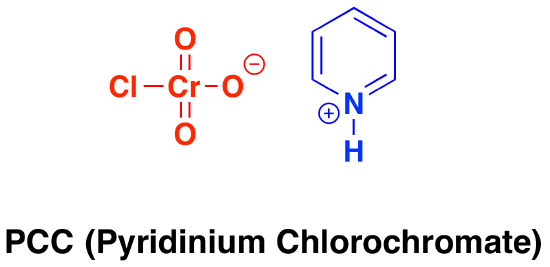
Oxidation by PCC (pyridinium chlorochromate)
- Page ID
- 15407
Pyridinium chlorochromate (PCC) is a milder version of chromic acid.
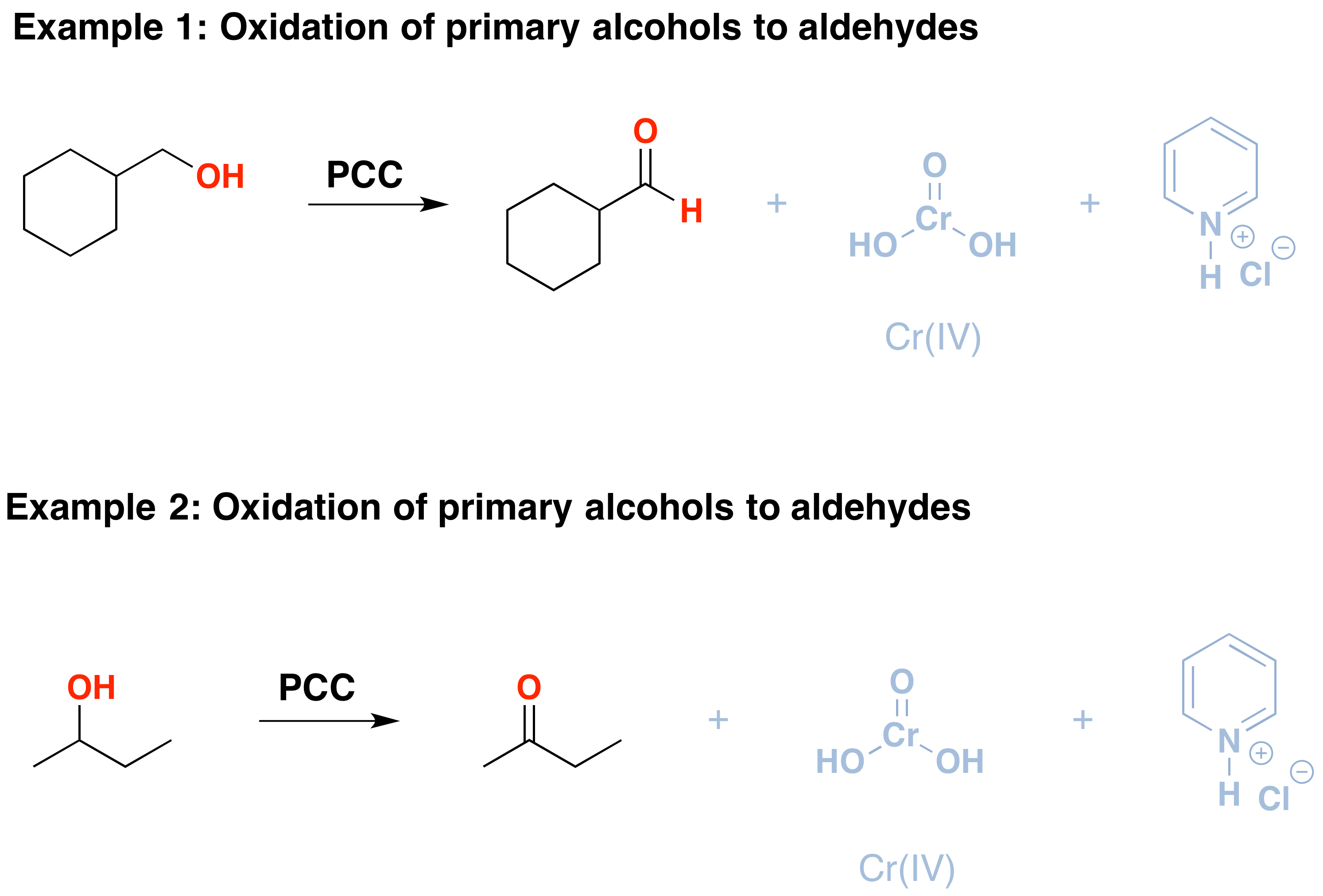
PCC oxidizes alcohols one rung up the oxidation ladder, from primary alcohols to aldehydes and from secondary alcohols to ketones. In contrast to chromic acid, PCC will not oxidize aldehydes to carboxylic acids. Similar to or the same as: \(CrO_3\) and pyridine (the Collins reagent) will also oxidize primary alcohols to aldehydes. Here are two examples of PCC in action.
- If you add one equivalent of PCC to either of these alcohols, the oxidized version will be produced. The byproducts (featured in grey) are Cr(IV) as well as pyridinium hydrochloride.
- One has to be careful with the amount of water present in the reaction. If water is present, it can add to the aldehyde to create the hydrate, which could be further oxidized by a second equivalent of PCC if it is present. This is not a concern with ketones because there is no H directly bonded to C.
How does it work? Oxidation reactions of this sort are actually a type of elimination reaction. The reaction starts with a carbon-oxygen single bond and results in a carbon-oxygen double bond. The elimination reaction can occur because of the good leaving group on the oxygen, namely the chromium, which will be displaced when the neighboring C-H bond is broken with a base.
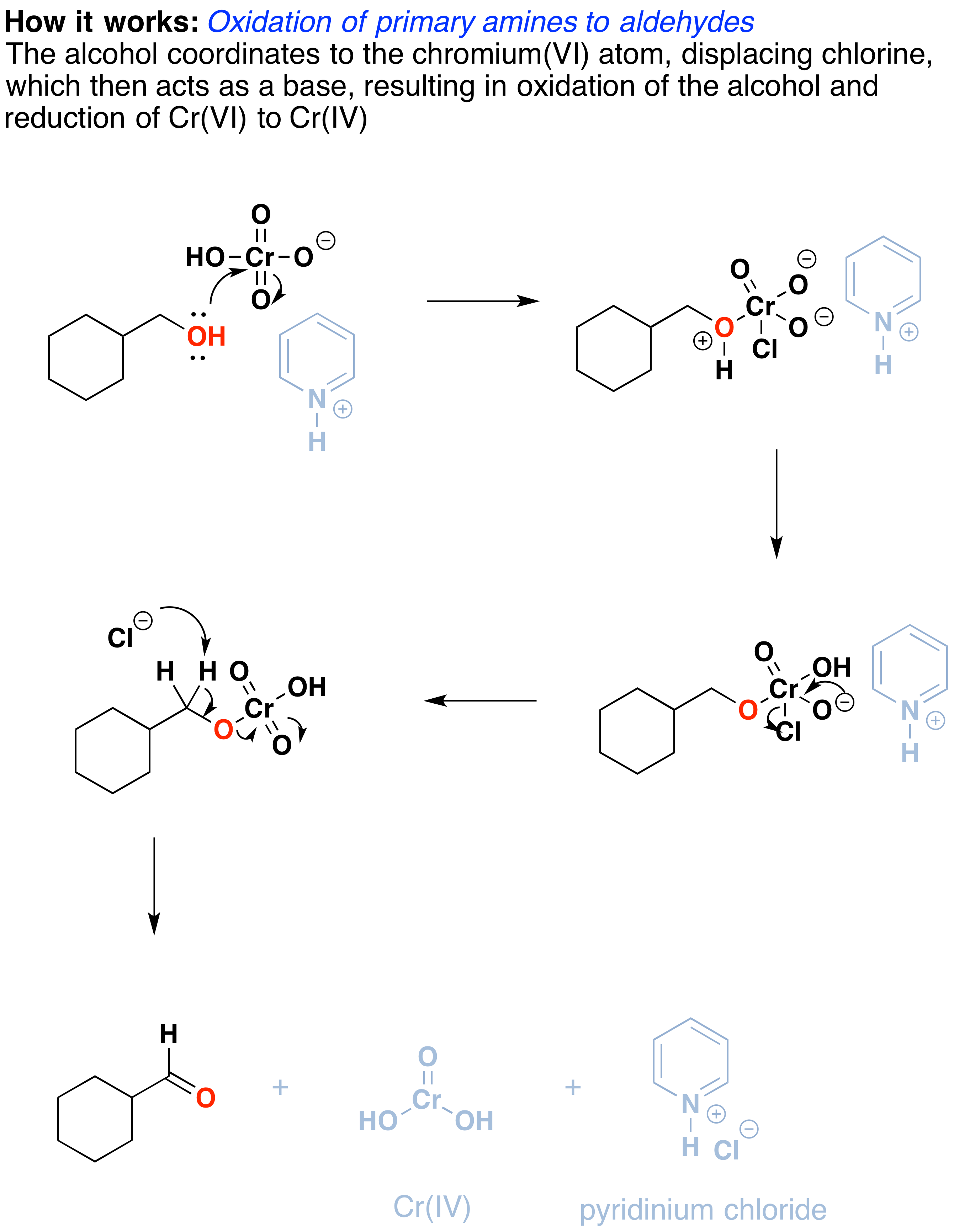
The first step is attack of oxygen on the chromium to form the Cr-O bond. Secondly, a proton on the (now positive) OH is transferred to one of the oxygens of the chromium, possibly through the intermediacy of the pyridinium salt. A chloride ion is then displaced in a reaction reminiscent of a 1,2 elimination reaction to form what is known as a chromate ester.
The C-O double bond is formed when a base removes the proton on the carbon adjacent to the oxygen. [aside: I've drawn the base as Cl(-) although there are certainly other species which could also act as bases here (such as an alcohol). It is also possible for pyridine to be used as the base here, although only very low concentrations of the deprotonated form will be present under these acidic conditions.] The electrons from the C-H bond move to form the C-O bond, and in the process, the O-Cr bond is broken, and Cr(VI) becomes Cr(IV) (drawn here as O=Cr(OH)2 ).
Real life notes: If you end up using PCC in the lab, do not forget to add molecular sieves or Celite or some other solid to the bottom of the flask because otherwise you get a nasty brown tar that is difficult to clean up. The toxicity and mess associated with chromium has spurred the development of other alternatives, such as TPAP, IBX, DMP, and a host of other reagents you generally do not typically learn about until grad school.
Contributors
James Ashenhurst (MasterOrganicChemistry.com)

