In-class Problem Set #2
- Page ID
- 72948
Before beginning this problem set, I spend about five minutes introducing peak broadening and reminding them about the ideal chromatographic peak. I also remind them of the concept of theoretical plates, the idea that we want a plate height that is as small as possible, and propose that we will spend the next section of the course examining the question: “What contributes to peak broadening and how can we reduce it?”
Longitudinal Diffusion Broadening
1. Consider a “band” of a compound in a chromatographic column. The band had the following concentration profile:
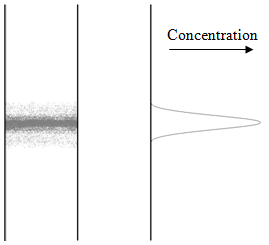
a) What would happen to this profile if the flow of the column were stopped and the column was allow to sit?
Students may be confused about the diagram that accompanies this question. Some students may be tempted to say that band will shrink, others might say it will spread out but not realize why, others may even think that since the flow has stopped it will stay exactly the same. After allowing the students some time to consider this question, summarize how diffusion will cause this peak to become broader.
b) Would this phenomena be more significant (i.e. happen faster) in a gas or a liquid?
Students should realize right away that diffusion occurs faster in a gas, but ask them how much faster. You may get answers ranging from ten times to ten million times faster.
c) Does this phenomenon contribute more to band broadening at higher or lower flow rates?
Most groups realize that a slower flow rate means that the compounds will have more time in the column and then more time for longitudinal diffusion to occur. Students should realize that this means that there is a greater contribution to peak broadening from longitudinal diffusion at lower flow rates. I summarize this conclusion, set up the concept of how we will be developing something known as the van Deemter equation that will eventually include terms for all of the different contributions to peak broadening, and then ask them:
Should the term for the contribution to peak broadening from longitudinal broadening be multiplied or divided by the mobile phase velocity.
Even though they understand that there is a greater contribution at lower flow rates, they may have trouble understanding how to incorporate that into the equation. With a few minutes time, each group arrives at the correct form of the expression to include in the equation.
Eddy Diffusion Broadening
Consider molecules flowing through a packed bed of particles.

a) Would different molecules have different path lengths as they passed through the bed?
Students should have no trouble with this question.
b) Is the difference in path length between the shortest and longest path dependent at all on the diameter of the particle? If so, which particles (smaller or larger) would lead to a greater difference?
Students will immediately acknowledge that there is a dependence on particle size, but may not know which will lead to a greater difference. Some may argue that with larger particles there are fewer paths that lead straight out of the column and with smaller particles there is the possibility of zigzagging all the way across the column. If anything over the years I have taught this course, more students think that smaller particles will be worse than larger particles. I encourage them to only consider realistic paths; with the flow pushing the molecule down the column, the molecule will not zigzag across the column. Eventually we get to the point of agreement that large particles lead to the greatest differences in path length.
c) Some packed columns exhibit channeling. What do you think is meant by this term?
Some students think that channeling has to do with the large interstitial regions resulting from using large particles. Other may recognize that it has something to do with improperly packed columns. Allow them just a few minutes to think about this and then discuss streamlined paths and the problem this introduces with regards to broadening of peaks.
d) Would channeling be more likely to occur with smaller or larger particles? In other words, which is more difficult to pack efficiently, larger or smaller particles? A column is packed efficiently when the particles are in a uniform bed with the minimum amount of voids.
A majority of the students will probably argue that it is harder to pack large particles efficiently. Remind them that efficient packing has to do with minimizing the voids; just because large particles result in large voids, it doesn’t mean that the voids can’t be minimized. Encourage them to imagine trying to pack large and small objects efficiently (fitting in the most number of particles possible in the allotted space implies the tightest and most efficient packing). How easy is it to get the smaller particles perfectly settled in relative to larger particles? It may take a while for the students to be convinced that smaller particles are harder to pack efficiently.
e) Do open tubular capillary columns exhibit eddy diffusion?
Students should have no trouble with this question. They should realize that having no particles means that there is no eddy diffusion.
f) Does this phenomenon exhibit any dependence on flow rate?
This is actually a highly debated question with conflicting data and representations in the literature (see text for a further discussion). Students may initially be tempted to say that there is no dependence on flow rate, which is consistent with the initial form of the van Deemter equation. They will argue that since eddy diffusion just has to do with the difference in path length, the time that it takes to travel those paths is insignificant. Encourage them to think about this question in more depth. Won’t a slower flow rate allow the molecules to sample more of the column? Won’t a faster flow rate cause molecules to be more likely to stay in one path or channel? Allow them to debate both arguments and then discuss how this is still a greatly debated topic. Introduce the eddy diffusion term and include it in the van Deemter equation.
Stationary Phase Mass Transport Broadening
Consider a compound that has distributed between the mobile and the stationary phase within a plate in a chromatographic column. The following diagram might represent the concentration distribution profiles in the two phases (note that the compound depicted has a preference for the mobile phase).
a) What would happen to these concentration profiles a brief instant of time later?
Allow students a few minutes to think about this problem. Some students may think that it will just stay the same. Some students may think that both concentration profiles will shift down. Remind them of the key difference between a mobile phase and a stationary phase. Encourage them to think about what would happen to molecules in the stationary phase at the top of the diagram once the molecules in the mobile phase have shifted down. What about the molecules in the mobile phase at the bottom of the diagram?
b) Will what happens in (a) contribute to band broadening? Explain.
Students should realize that this will contribute to peak broadening because the overall Gaussian distribution becomes wider. Discuss how molecules must spend a finite amount of time in the stationary phase. This is a good time to discuss that the lag in mass transport that effectively traps molecules in the stationary phase for a finite amount of time means that a chromatographic column is constantly at disequilibrium (even though we often present it as if the compound equilibrates over the section of the column where it is distributing between the two phases).
c) Does the contribution of this phenomenon to band broadening exhibit a dependence on flow rate? If so, are there any troublesome aspects to this dependence?
Students should realize that the faster the flow rate, the more broadening there will be. You may ask them to draw two profiles for the distribution in the mobile phase a brief instant of time later for one flow rate and then double that flow rate. Once students realize the correct answer, spend a few minutes summarizing the conclusions and talking about how with slower flow rates the system is able to reach a state that is closer to equilibrium. Introduce the stationary phase mass transport term for the van Deemter equation. Allow students some time to realize that they now have one term in the equation that recommends use of a faster flow rate and one term that recommends use of a slower flow rate to optimize efficiency.
d) What happens to this effect as the stationary phase coating is made thicker?
Students should realize right away that with a thicker coating, it will take longer for particles to diffuse out of the stationary phase and therefore broadening will be increased. At this point, I then ask them:
How could you create a packed column with a thinner coating of stationary phase?
Students usually decide to apply a lower percent coating, but talk about the disadvantages (reduced capacity) of doing that. Ask them to think of a method that retains the same percent loading but does not decrease the capacity. The students should eventually think of using smaller particles in order to keep the same weight of the liquid phase and solid support but increase the surface area. Discuss some of the drawbacks to using smaller particles (need to take more care in coating to insure uniform surface coverage, more care in packing to avoid channeling).
e) Capillary GC columns have very thin coatings. Describe one advantage of these columns.
After completing part (d), students realize that a thinner coating will minimize the broadening due to stationary phase mass transport. I usually spend a few minutes talking about how liquid coatings are applied to capillary columns. This is also a good time to talk about the history of capillary gas chromatography including how glass capillary columns used to be made. Also discuss the use of fused silica capillary columns and how they compare to regular glass. It would also be a good idea to mention the low capacity of these types of columns and the use of a split injection system. Talk about the role that fiber optics played in the development of capillary gas chromatography.
f) Compare the effect of this phenomenon on a uniform versus non-uniform stationary phase coating.
The groups are usually quick to realize that a non-uniform coating will be undesirable. I summarize this, spend a few minutes talking about the methods for coating particles, and remind them of the deleterious effect if the coating process leads to regions of exposed surface that will result in adsorption.
g) Is this effect of more concern in gas or liquid chromatography?
Allow the students a few minutes to consider this question. The students often focus on the difference in the mobile phases of gas and liquid chromatography, and assume that since gases diffuse faster than liquids, are tempted to say that this is more of a concern with liquid chromatography. Remind them that stationary phase mass transport broadening just has to do with the amount of time a molecule stays in the stationary phase. Once a molecule has entered the stationary phase, it doesn’t matter whether the mobile phase is a gas or a liquid. In our current understanding, the stationary phases in gas and liquid chromatography are both liquids. Challenge them to think of some other key differences between gas and liquid chromatography. Ask them what temperature a gas chromatograph oven is usually at compared to a liquid chromatography column. With this information, you can ask them the effect that the different temperatures of gas and liquid chromatographic columns will have on diffusion rates. They should now see that stationary phase mass transport broadening is likely to be more of a concern in liquid chromatography, but not as much as they originally thought when they only considered diffusion rates within the mobile phase.
At this point, I discuss the explosion of gas chromatography as an analysis method in the 1950s-70s and how that led to efforts to volatilize non-volatile compounds.
I also introduce Calvin Giddings and his paper titled, Liquid Chromatography with Operating Conditions Analogous to Those of Gas Chromatography, which concludes that ultra-small particles will be needed if LC is to have an efficiency comparable to GC (although we have not yet discussed mobile phase mass transport broadening).
What issues might there be in coating ultra-small particles with a very thin liquid coating?
What issues arise trying to keep a liquid coating on ultra-small particles when a liquid is the mobile phase?
I usually pose these two questions to the entire class rather than the groups and solicit individual student responses. We then discuss issues such as coating uniformity and degradation of the liquid coating due to physical removal from the solid support by the flowing liquid and because the liquid coating and liquid mobile phase will still have some slight solubility in each other even if they are regarded as immiscible (while oil and water don’t mix, they actually do to some small extent). I then spend some time talking about the development of bonded phases for liquid chromatography (especially C-18 phases) and the significance of this discovery to the field of liquid chromatography.
Mobile Phase Mass Transport Broadening
Consider a capillary column as shown below.
The dot represents a molecule that has just left the stationary phase is about to diffuse across the mobile phase and re-encounter the stationary phase on the other side of the column.
a) Draw a line representing the path of this molecule.
Most students realize right away to draw a line that traverses from one side of the channel to the other. Some need prompting (usually to look at what another student in the group has drawn).
b) What would this path look like if the flow rate were doubled?
Students recognize that this would result in the molecule being pulled further down the column before encountering the stationary phase again.
c) Is it important for the molecule to encounter the stationary phase? Think about a situation in which the flow was so fast that the molecule never re-encountered the stationary phase.
Students realize that without a molecule encountering the stationary phase, there is no separation and therefore no chromatography. Therefore the contribution of mobile phase mass transport broadening is more significant at higher flow rates.
d) If using capillary columns, what does this suggest about the desirable diameter for such a column?
Students realize right away that the narrower the column the better.
e) Is this phenomenon worse in gas or liquid chromatography?
Having fully developed the effect for the stationary phase, they realize right away that the much slower diffusion in liquids means that mobile phase mass transport broadening is much more significant in liquid chromatography.
f) How does the contribution to band broadening depend on flow rate?
Again, they usually realize it is exactly analogous to stationary phase mass transport broadening.
g) Would this effect be observed in a packed column? If so, how?
Generally, students realize immediately that the interstitial volume in a packed column is analogous to the open space in a capillary column.
h) How could you reduce this effect in a packed column?
Students usually immediately propose using smaller particles.
i) Say something about the length of a column needed if you undertake what you have suggested in (h), assuming that you have maintained a constant thickness of stationary phase.
I first point out that this is exactly the situation that occurs with a bonded phase liquid chromatographic column, where the thickness is set by the nature of the bonded C-18 groups and does not depend on the particle size they are bonded to. Students immediately realize that the smaller particle has a larger surface area, and then usually soon realize that this means that the capacity of a bonded phase LC column with smaller particles is greater than that with larger particles. With greater capacity they realize that, when using smaller particles in LC, you can maintain the same capacity by using a shorter column. When asked what else they will observe with a shorter column, most immediately realize that it will take less time to run the chromatogram.
I then spend a few minutes discussing plots of h versus flow rate and talk about the important differences between the plots for liquid chromatography and gas chromatography. Talk about the optimal flow rate and realistic flow rates.


