Detection Methods
- Page ID
- 71909
As mentioned previously, highly sensitive detection methods were developed for gas chromatography in the 1950s and 1960s that facilitated its use as an analytical method.
One of the most useful detection methods is known as the flame ionization detector (FID). The flame ionization detector is highly sensitive and involves burning the sample in an air-hydrogen flame. Molecules with a CH bond will form CH+ ions in the flame, and these are measured using a negatively charged collector above the flame. When these positive ions strike the negatively charged collector, a current proportional to the amount of ions is measured. This detector is essentially universal, as it is able to measure all organic compounds. It is not sensitive toward compounds like water and carbon dioxide, which is advantageous.
Another important detector in the development of gas chromatography is the thermal conductivity detector. This is a universal detector, but not nearly as sensitive as the FID. A resistance circuit called a Wheatstone bridge is used. The bridge has two halves, and the resistance of these halves is compared. Instruments with a thermal conductivity detector need two matched columns. One column has only carrier gas flowing through it. The other has carrier gas and sample. Changes in the thermal conductivity of the gas change the resistance reading over the Wheatstone bridge. Some representative values for themal conductivities of gases are listed below. The value for butane is representative of most organic compounds. Since we need to measure a difference in thermal conductivity, we need a carrier gas with as different a thermal conductivity as possible from most organic compounds. This would suggest the use of hydrogen gas as a carrier gas, however hydrogen is too much of an explosion risk for this purpose. Helium is therefore the carrier gas of choice. Thermal conductivity detectors tend to be used on less expensive pieces of equipment like the gas chromatographs used for the analysis of the purity of compounds prepared in organic chemistry instructional labs.
\(\begin{align}
\ce{&Thermal\: conductivity\: values: &&Hydrogen &471\\
& &&Helium &376\\
& &&Nitrogen &66\\
& &&\mathit{n}-Butane &43}
\end{align}\)
One of the most important gas chromatographic detectors ever developed was the electron capture detector. This device, which is illustrated in Figure 73, was invented by James Lovelock. The device has a radioactive foil (usually containing tritium or radioactive nickel) that emits beta particles. Beta particles are high-energy electrons emitted by a nuclear decay process (a neutron decays into a beta particle and a proton). The opposite side of the device is positively charged so the beta particles stream across the tube. As the high-energy beta particles strike carrier gas molecules (usually either nitrogen or an argon/methane mixture), they create a cascade of lower energy electrons, which generate a current that can be measured. If we had a compound coming out of the gas chromatograph that could capture some of these low-energy electrons, the current would drop. The drop in current can be related to the concentration of electron capturing compound in the sample. What is especially impressive about this detector is the level of sensitivity that can be achieved for certain classes of compounds.
What types of compounds ought to have the ability to capture electrons? Remembering back to your knowledge of periodic properties, you should predict that halogen-containing compounds (compounds with a chlorine, fluorine, or bromine atom) ought to be very effective at capturing electrons. What are especially significant are the classes of compounds that contain halogens. These include the following (with their respective application or concern):
- Chlorofluorocarbons (or freons) – ozone layer destruction
- Chloroform – potential carcinogen formed in drinking water from water chlorination
- Dioxin – by-product of combustion and certain industrial processes
- Chlorinated pesticides such as DDT
- Polychlorinated biphenyls – used in electronic devices (transformers)
These are only some examples, but they form an extensive array of compounds of environmental significance. The invention of the electron capture detector facilitated the discovery that freons were making their way to the stratosphere and leading to the destruction of the ozone layer. Without this detector, this discovery would have been delayed by many years.
Mass spectrometric detection is especially important because it provides information that can be used for compound identification. Most gas chromatograph-mass spectrometers (GC-MS) use fused silica capillary columns, and the column effluent can go straight into the mass spectrometer. What happens in the mass spectrometer is that the column effluent is bombarded with a high-energy beam of electrons. As these electrons strike the sample, they generate ions. These can be anions or cations, but most mass spectrometers are designed to draw off only the cations for analysis. Usually almost all the cations have a +1 charge. If we consider the molecule acetophenone, we can illustrate what will take place (three functional groups within the molecule are labeled A, B, and C).
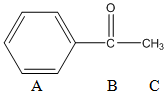
Acetophenone
If an electron struck this molecule and knocked out one other electron, we would have the entire molecule with a +1 charge. This ion is referred to as the molecular ion, and we would show this as ABC+. It’s usually very desirable to have a molecular ion in the spectrum. The other valuable observation in mass spectrometry when you use an electron beam for ionization is that you get fragment ions as well. Possible fragments we could get from acetophenone are listed in Table 5, along with their masses. Actually, what is really measured is the mass-to-charge ratio, denoted as m/e. If all of the ions have a charge of +1, the mass equals the mass-to-charge ratio.
Table 5. Possible fragment ions from acetophenone using A, B and C to denote pieces of the molecule.
| Ion | m/e |
|---|---|
| ABC+ | 120 |
| AB+ | 105 |
| AC+ | 92 |
| A+ | 77 |
| BC+ | 43 |
| B+ | 28 |
| C+ | 15 |
A good question to ask is whether we would ever get AC+ as a fragment? This would require an intramolecular reaction to take place. This does happen with some molecules, but not the one we are using as an example. Similarly, it might be very unlikely to get a B+ ion, which would require the loss of two groups. If you were to examine a book on reaction mechanisms that occur in a mass spectrometer, you would notice that it would look very similar to an organic chemistry textbook. The two things we observe is that different fragments have different weights, but they also have different intensities. For example, if a molecule has a good leaving group, it will tend to leave in the mass spectrometer. Depending on which fragment is more stable as a positive ion, the relative intensity of the two ions will vary. The most intense ion is given a value of 100 and the intensity of each other ion is reported relative to it. A possible mass spectrum for acetophenone is provided in Figure 74. It is worth noting that there will be many more ions in the spectrum than these. For one thing, isotope effects show up. It turns out that 1% of all carbon is carbon-13 so one out of every hundred molecules weighs one more (meaning that we will see a small peak at m/e = 121). Note that the mass spectrometer does not measure an average molecular weight based on isotopic abundance, but the exact weight of each ion. For another thing, the molecule might lose a hydrogen atom and show a fragment with a mass of 119.
Notice how we could use these masses to determine a possible molecular formula for the compound, and the masses of fragments to determine possible groups that are found in the molecule. The compound below (2-phenylethanal) has the exact same molecular weight (120) as acetophenone, but you might appreciate that it would probably have a different mass spectrum.
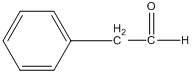
2-phenylethanal
Mass spectra are relatively difficult to interpret and to assign an unequivocal structure to. What is usually done instead is that the mass spectrometers with a GC-MS come with a library of spectra of known compounds. The computer will compare your measured mass spectrum to those in the library and report the ten best matches. The top match does not confirm the assignment. To do that, you would need an authentic sample of that compound and would have to show that it has the same retention time and mass spectrum on your instrument. With those two matches, you can be assured that you have identified the compound. GC-MS is commonly used for drug testing at sports events like the Olympics.


