1.9: Other Single Crystal Surfaces
- Page ID
- 25362
Although some of the more common metallic surface structures have been discussed in previous sections (1.2-1.4), there are many other types of single crystal surface which may be prepared and studied. These include
- high index surfaces of metals
- single crystal surfaces of compounds
These will not be covered in any depth, but a few illustrative examples are given below to give you a flavor of the additional complexity involved when considering such surfaces.
High Index Surfaces of Metals
High index surfaces are those for which one or more of the Miller Indices are relatively large numbers. The most commonly studied surfaces of this type are vicinal surfaces which are cut at a relatively small angle to one of the low index surfaces. The ideal surfaces can then be considered to consist of terraces which have an atomic arrangement identical with the corresponding low index surface, separated by monatomic steps (steps which are a single atom high).
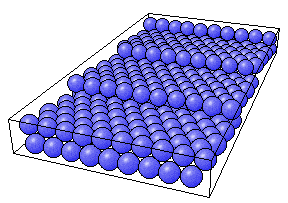
Perspective view of the fcc(775) surface
As seen above, the ideal fcc(775) surface has a regular array of such steps and these steps are both straight and parallel to one another.
What is the coordination number of a step atom on this surface ?
- Answer
-
The coordination number of the atoms at the steps is 7
Rationale: Each step atom has four nearest neighbors in the surface layer of terrace atoms which terminates at the step, and another three in the layer immediately below ; a total of 7. This contrasts with the CN of surface atoms on the terraces which is 9.
By contrast a surface for which all the Miller indices differ must not only exhibit steps but must also contain kinks in the steps. An example of such a surface is the fcc(10.8.7) surface - the ideal geometry of which is shown below.

Perspective view of the fcc(10.8.7) surface
What is the lowest coordination number exhibited by any of the atoms on this surface?
- Answer
-
The lowest coordination number is 6 which is that exhibited by atoms at the kinks in the steps.
Rationale: The lowest coordination number is exhibited by atoms "on the outside" of the kinks in the steps. Such atoms have only three nearest neighbors in the surface layer of terrace atoms which terminates at the step, and another three in the layer immediately below ; a total of 6. This contrasts with the surface atoms on the terraces which have a coordination number of 9 and the normal step atoms which have a coordination number of 7.
Real vicinal surfaces do not, of course, exhibit the completely regular array of steps and kinks illustrated for the ideal surface structures, but they do exhibit this type of step and terrace morphology. The special adsorption sites available adjacent to the steps are widely recognized to have markedly different characteristics to those available on the terraces and may thus have an important role in certain types of catalytic reaction.
Single Crystal Surfaces of Compounds
The ideal surface structures of the low index planes of compound materials can be easily deduced from the bulk structures in exactly the same way as can be done for the basic metal structures. For example, the NaCl(100) surface that would be expected from the bulk structure is shown below:
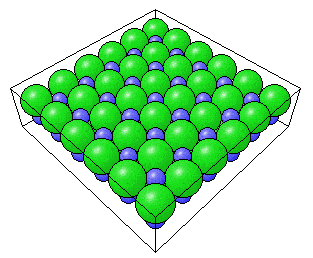
Perspective view of the NaCl(100) surface
In addition to the relaxation and reconstruction exhibited by elemental surfaces, the surfaces of compounds may also show deviations from the bulk stoichiometry due to surface localised reactions (e.g. surface reduction) and/or surface segregation of one or more components of the material.


