5.14: Model Diffraction Pattern for Napthalene
- Page ID
- 150539
Ten holes in a two-dimensional mask are used to model the diffraction pattern for napthalene. It is assumed that only the carbon atoms scatter radiation.
Establish mask geometry:
\[ \begin{matrix} R= 2 & m = 1 .. 6 & \Theta_m = \frac{2 \pi m}{6} & x_m = R \sin ( \Theta_m ) - \sqrt{3} & y_m = R \cos ( \Theta_m) \\ ~ & m = 7 .. 10 & \Theta_m = \frac{2 \pi (m - 0.5)}{6} & x_m = R \sin ( \Theta_m) + \sqrt{3} & y_m = R \cos ( \Theta_m - \frac{ \pi}{ \sqrt{3}} ) \end{matrix} \nonumber \]
Fourier transform of position wave function (mask geometry) into the momentum representation. Initially the carbon atoms are considered to be point scatterers.
\[ \begin{matrix} m = 1 .. 10 & \Phi (p_x,~p_y) = \frac{1}{2 \pi d \sqrt{10}} \left[ \sum_{m = 1} \left( \int_{x_m - \frac{d}{2}}^{ x_m + \frac{d}{2}} exp(-i p_x x) dx \int_{y_m - \frac{d}{2}}^{y_m + \frac{d}{2}} exp (-i p_y y) dy \right) \right] \end{matrix} \nonumber \]
Display mask geometry and diffraction pattern:
\[ \begin{matrix} N = 100 & \Delta p = 15 & j = 0 .. N & k = 0 .. N & px_j = - \Delta + \frac{2 \Delta p j}{N} & py_k = - \Delta p + \frac{2 \Delta p k}{N} \end{matrix} \nonumber \]
\[ \text{Diffraction pattern}_{j,~k} = \left( \left| \Phi ( px_j,~py_k ) \right| \right)^2 \nonumber \]

This calculation illustrates the uncertainty principle. There is no attenuation in the diffraction pattern (momentum distribution) because there is no uncertainty in the carbon atom positions.
Now the diffraction pattern is recalculated assuming an arbitrary finite dimension for the carbon atoms.
\[ \begin{matrix} r = .15 & \Phi (p_x,~p_y) = \frac{1}{2 \pi d \sqrt{10}} \left[ \sum_{m = 1} \left( \int_{x_m - r}^{ x_m + r} exp(-i p_x x) dx \int_{y_m - r}^{y_m + r} exp (-i p_y y) dy \right) \right] \end{matrix} \nonumber \]
\[ \text{Diffraction pattern}_{j,~k} = \left( \left| \Phi ( px_j,~py_k ) \right| \right)^2 \nonumber \]
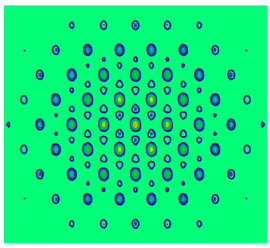
There is attenuation in the momentum distribution (less uncertainty) because there is more uncertainty in the carbon atom positions.

