5.6: Resolving Kinetics- Slow Methods
- Page ID
- 9253
These methods are applicable to reactions that are not excessively fast, typically requiring a few minutes or hours to run to completion. They were about the only methods available before 1950. Most first-year laboratory courses students will include at least one experiment based on one of these methods.
Optical methods
Light absorption is perhaps the most widely-used technique. If either a reactant or a product is colored, the reaction is easily followed by recording the change in transmission of an appropriate wavelength after the reaction is started. When a beam of light passes through a solution containing a colored substance, the fraction that is absorbed is directly proportional to the concentration of that substance and to the length of the light's path through the solution. The latter can be controlled by employing a cell or cuvette having a fixed path length.
If I0 is the intensity of the light incident on the cell and I is the intensity that emerges on the other side, then the percent absorption is just 100 × I / I0. Because a limited 1-100 scale of light absorption is often inadequate to express the many orders of magnitude frequently encountered, a logarithmic term optical density is often employed. The relation between this, the cell path length, concentration, and innate absorption ability of the colored substance is expressed by Beer's Law.
The simplest absorbance measuring device is a colorimeter in which a beam of white light from an incandescent lamp is passed through a cell (often just a test tube) and onto a photodetector whose electrical output is directly proportional to the light intensity. Before beginning the experiment, a zeroing control sets the meter to zero when the light path is blocked off, and a sensitivity control sets it to 100 when a cell containing an uncolored solution (a "blank") is inserted.
The sensitivity and selectivity of such an arrangement is greatly enhanced by adjusting the wavelength of the light to match the absorption spectrum of the substance being measured. Thus if the substance has a yellow color, it is because blue light is being absorbed, so a blue color filter is placed in the light path.
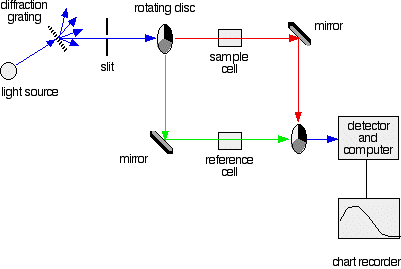
More sophisticated absorption spectrophotometers employ two cells, one for the sample and another reference cell for the blank. They also allow one to select the particular wavelength range that is most strongly absorbed by the substance under investigation, which can often extend into the near-ultraviolet region.
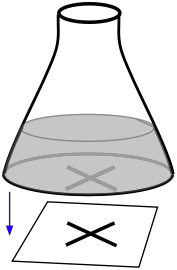
Light scattering measurements (made with a nephthelometer) can be useful for reactions that lead to the formation a fine precipitate. A very simple student laboratory experiment of this kind can be carried out by placing a conical flask containing the reaction mixture on top of a marked piece of paper. The effects of changing the temperature or reactant concentrations can be made by observing how long it takes for precipitate formation to obscure the mark on the paper.
Other optical methods such a fluorescence and polarimetry (measurement of the degree to which a solution rotates the plane of polarized light) are also employed when applicable.
Other methods
- Measurement of gases: The classical method of following reactions that produce changes in the number of moles of gases is to observe changes in pressure or volume. An alternative method for following the loss of a gas is to place the reaction container on an electronic balance and monitor the loss in weight. This is generally less accurate than pressure measurements, but is sometimes used in student experiments.
- pH measurements: Many reactions yield or consume hydrogen ions and are conveniently followed by means of a pH meter.
- Electrical Conductance: Reactions that yield or consume ionic substances are often studied by measuring the electrical conductance of the solution. Conductimetry is usually carried out by balancing the solution conductance with a known resistance in a bridge arrangement. An audio-frequency alternating current is used in order to avoid electrolysis.


