2.58: The Wigner Distribution for the 3s State of the 1D Hydrogen Atom
- Page ID
- 158672
This tutorial presents three pictures of the 3s state of the one‐dimensional hydrogen atom using its position, momentum and phase‐space representations.
The energy operator for the one‐dimensional hydrogen atom in atomic units is:
\[ \frac{-1}{2} \frac{d^2}{dx^2} \blacksquare - \frac{1}{x} \blacksquare \nonumber \]
The position 3s wave function is:
\[ \begin{matrix} \Psi (x) = \frac{2}{243} \sqrt{3} \text{x} \left( 27-18x + 2x^2 \right) \text{exp} \left( - \frac{x}{3} \right) & \int_0^{ \infty} \Psi (x)^2 dx = 1 \end{matrix} \nonumber \]
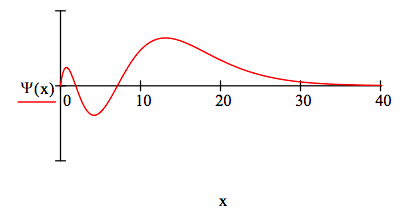
The 3s energy is -0.065 Eh.
\[ \Phi (p) = \frac{1}{ \sqrt{2 \pi}} \int_0^{ \infty} \text{exp(-i p x)} \Psi (x) \text{dx} \rightarrow \left( -2^{ \frac{1}{2}} \right) 3^{ \frac{1}{2}} \frac{9p^2 + 6ip - 1}{(3ip + 1)^4 \pi^{ \frac{1}{2}}} \nonumber \]
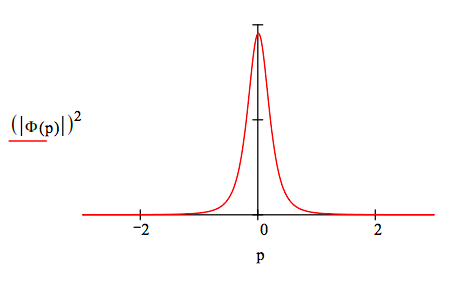
The Wigner function (phase‐space representation) for the hydrogen atom 3s state is generated using the momentum wave function.
\[ \text{W(x, p)} = \frac{1}{2 \pi} \int_{- \infty}^{ \infty} \overline{ \Phi \left( \text{p} + \frac{}{2} \right)} \text{exp(-i s x)} \Phi \left( \text{p} - \frac{ \text{s}}{2} \right) \text{ds} \nonumber \]
The Wigner distribution is displayed graphically.
\[ \begin{matrix} N = 100 & i = 0 .. N & x_i = \frac{30i}{N} & j = 0 .. N & p_j = -2 + \frac{4j}{N} & \text{Wigner}_{i,~j} = \text{W} \left( x_i,~ p_j \right) \end{matrix} \nonumber \]
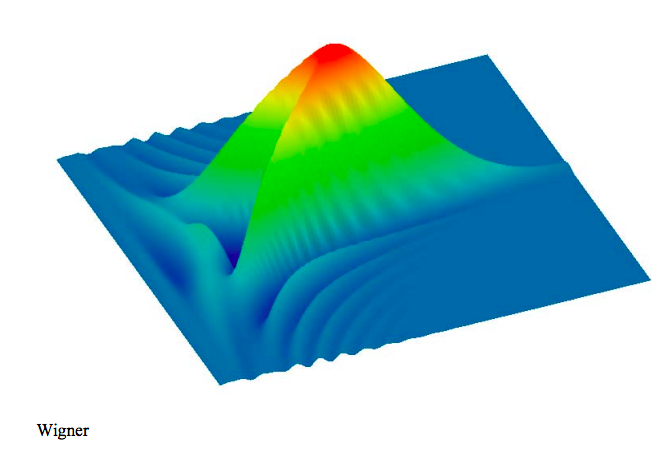
Just as for the 2s state, the Wigner distribution for the 3s state takes on negative values.

