2.10: Electronic Structure and the Superposition Principle
- Page ID
- 154884
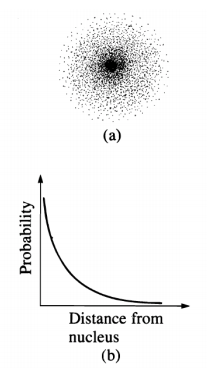
Electrons in atoms or molecules are characterized by their entire distributions, called wave functions or orbitals, rather than by instantaneous positions and velocities: an electron may be considered always to be, with appropriate probability, at all points of its distribution, which does not vary with time. (F. E. Harris)
For example, the hydrogen atom electron is in a stationary state which is a weighted superposition of all possible distances from the nucleus. The electron is not orbiting the nucleus; it does not execute a classical trajectory during its interaction with the nucleus.
From the quantum mechanical perspective, to measure the position of an electron is not to find out where it is, but to cause it to be somewhere. (Louisa Gilder)

