4.8: Statistics for Molecular Speeds
- Page ID
- 151991
Expected values for several quantities can be calculated from the Maxwell-Boltzmann probability density function. The required definite integrals are tabulated in Appendix D.
The most probable speed, \(v_{mp}\), is the speed at which the Maxwell-Boltzmann equation takes on its maximum value. At this speed, we have
\[ \begin{align*} 0&=\frac{d}{dv}\left(\frac{df\left(v\right)}{dv}\right)=\frac{d}{dv}\left[4\pi {\left(\frac{m}{2\pi kT}\right)}^{3/2}v^2\exp\left(\frac{-mv^2}{2kT}\right)\right] \\[4pt] &=\left[4\pi {\left(\frac{m}{2\pi kT}\right)}^{3/2}\exp\left(\frac{-mv^2}{2kT}\right)\right]\left[2v-\frac{mv^3}{kT}\right] \end{align*} \]
from which
\[v_{mp}=\sqrt{\frac{2kT}{m}}\approx 1.414\sqrt{\frac{kT}{m}} \nonumber \]
The average speed, \(\overline{v}\) or \(\left\langle v\right\rangle\), is the expected value of the scalar velocity (\(g\left(v\right)=v\)). We find
\[\overline{v}=\left\langle v\right\rangle =\int^{\infty }_0{4\pi {\left(\frac{m}{2\pi kT}\right)}^{3/2}v^3exp\left(\frac{-mv^2}{2kT}\right)}dv=\sqrt{\frac{8kT}{\pi m}}\approx 1.596\sqrt{\frac{kT}{m}} \nonumber \]
The mean-square speed, \(\overline{v^2}\) or \(\left\langle v^2\right\rangle\), is the expected value of the velocity squared (\(g\left(v\right)=v^2\)):
\[\overline{v^2}=\left\langle v^2\right\rangle =\int^{\infty }_0{4\pi {\left(\frac{m}{2\pi kT}\right)}^{3/2}v^4exp\left(\frac{-mv^2}{2kT}\right)}dv=\frac{3kT}{m} \nonumber \]
and the root mean-square speed, \(v_{rms}\), is
\[v_{rms}=\sqrt{\left\langle v^2\right\rangle }=\sqrt{\frac{3kT}{m}}\approx 1.732\sqrt{\frac{kT}{m}} \nonumber \]
Example \(\PageIndex{1}\)
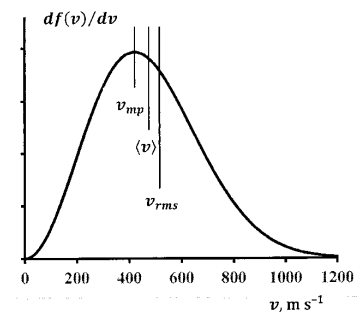
Figure 6 shows the velocity distribution 300 K for nitrogen molecules at 300 K.
Solution
Finally, let us find the variance of the velocity; that is, the expected value of \(\left(v-\left\langle v\right\rangle \right)^2\):
\({\text { variance }(v)=\sigma_{v}^{2}} \)
\[\begin{align*} &=\int_{0}^{\infty}(v-\langle v\rangle)^{2}\left(\frac{d f(v)}{d v}\right) d v \\ &=\int_{0}^{\infty} v^{2}\left(\frac{d f}{d v}\right) d v-2\langle v\rangle \int_{0}^{\infty} v\left(\frac{d f}{d v}\right) d v+\langle v\rangle^{2} \int_{0}^{\infty}\left(\frac{d f}{d v}\right) \\ &=\left\langle v^{2}\right\rangle- 2\langle v\rangle\langle v\rangle+\langle v\rangle^{2} \\ &=\left\langle v^{2}\right\rangle-\langle v\rangle^{2} \end{align*} \]
For \(N_2\) at \(300\) K, we calculate:
\[v_{mp}\ =422\ \mathrm{m\ }{\mathrm{s}}^{\mathrm{-1}} \nonumber \]
\[\left\langle v\right\rangle =\overline{v}=476\ \mathrm{m\ }{\mathrm{s}}^{\mathrm{-1}} \nonumber \]
\[v_{rms}=517\ \mathrm{m\ }{\mathrm{s}}^{\mathrm{-1}} \nonumber \]
\[ \text{Variance} \left(v\right)=\sigma^2_v=40.23\times {10}^{-3}\ \mathrm{m\ }{\mathrm{s}}^{\mathrm{-1}} \nonumber \]
\[\sigma_v=201\ \mathrm{m\ }{\mathrm{s}}^{\mathrm{-1}} \nonumber \]


