9.9.9E: Electron Affinity
- Page ID
- 64787
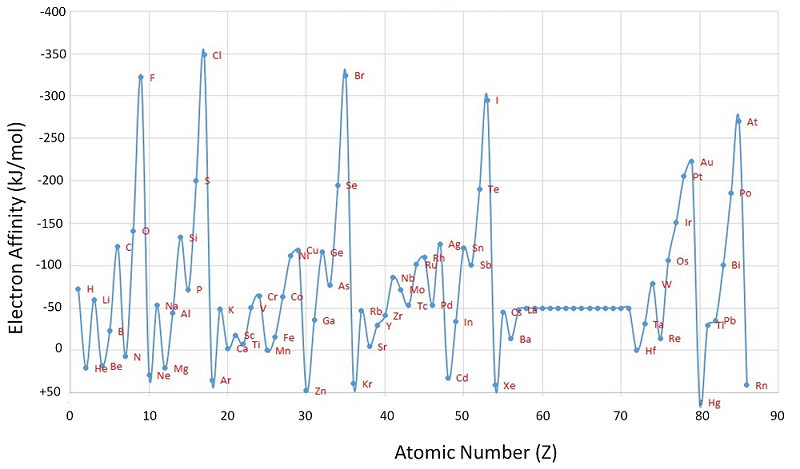
The inverse of ionization, i.e. bringing an electron from infinity to occupy the lowest-energy vacancy in an atomic orbital, produces an energy change called the electron affinity.
\[ A + e^- \rightarrow A^- \label{9-69}\]
with
\[\Delta E = E_f - E_i = EA \label {9-70}\]
Negative electron affinities, meaning that the above reaction is exothermic, are found for neutral atoms that are actually higher in energy than the corresponding anion, such as the halogens. The electron affinities of elements, just like their ionization potentials, are important in understanding their chemical reactivity and bonding characteristics. The electron affinity of a neutral atom is actually just the negative of an ionization energy for an anion (i.e, the reverse reaction for Equation \(\ref{9-69}\)).
\[A^- \rightarrow A + e^-\]
with
\[\Delta E = E_f -E_i = IP_A = - EA_A \label {9-71}\]
As above, the best values for electron affinities are obtained by calculating the difference in total binding energy between an anion and a neutral atom.