8.8: Coupling of Angular Momentum and Spectroscopic Term Symbols
- Page ID
- 4520
Around 1930 several spectroscopists using high resolution instruments found that lines in the hydrogen atom spectrum actually are not single lines but they are multiplets as shown for an isotopic mixture of hydrogen,\((H^1_{\alpha})\) and deuterium, (\(H^2_{\alpha}\)) in Figure \(\PageIndex{1}\). A multiplet consists of two or more closely spaced lines. Two lines together form a doublet, three a triplet, etc. Multiplets also are called fine structure. The term fine structure means the lines are spaced close together, i.e. finely spaced. Such fine structure also was found in spectra of one-electron ions such as He+.

You should recall that the \(H^1_{\alpha}\) line in the Balmer series at 656.279 nm was understood as resulting from a single transition of an electron from the n = 3 energy level to the n = 2 level. The observation of fine structure revealed that an orbital energy level diagram does not completely describe the energy levels of atoms. This fine structure also provided key evidence at the time for the existence of electron spin, which was used not only to give a qualitative explanation for the multiplets but also to furnish highly accurate calculations of the multiplet splittings.
Spin-Orbit Coupling
Specifying the orbital configuration of an atom does not uniquely identify the electronic state of the atom because the orbital angular momentum, the spin angular momentum, and the total angular momentum are not precisely specified. For example in the hydrogen 2p1 configuration, the electron can be in any of the three p-orbitals, \(m_l\) = +1, 0, and –1, and have spins with \(m_s\) = +1/2 or –1/2. Thus, there are 3 times 2 different possibilities or states. Also, the orbital and spin angular momentum of the electrons combine in multiple ways to produce angular momentum vectors that are characteristic of the entire atom not just individual electrons, and these different combinations can have different energies. This coupling of orbital and spin angular momentum occurs because both the electron spin and orbital motion produce magnetic dipole moments. As we have seen previously, the relationship between the angular momentum and the magnetic moment is given by the gyromagnetic ratio. These magnetic dipoles interact just like two tiny bar magnets attracting and repelling each other. This interaction is called spin-orbit interaction. The interaction energy is proportional to the scalar product of the magnetic dipole moments, which are proportional to the angular momentum vectors.
\[E_{s-o} = \lambda S \cdot L \label{8.8.1}\]
\[\hat {H} _{s-o} = \lambda \hat {S} \cdot \hat {L} \label {8.8.2}\]
where \(\lambda\) represents the constant of proportionality and is called the spin-orbit coupling constant. The spin-orbit interaction couples the spin motion and orbital motion of all the electrons together. This coupling means that exact wavefunctions are not eigenfunctions of the spin and orbital angular momentum operators separately. Rather the total angular momentum J = L+S, the vector sum of the spin and orbital angular momentum, is required to be coupled for a completely accurate description of the system. Trying to describe the coupled system in terms of spin and orbital angular momentum separately is analogous to trying to describe the positions of two coupled bar magnets independently. It cannot be done; their interaction must be taken into account.
We need to be able to identify the electronic states that result from a given electron configuration and determine their relative energies. An electronic state of an atom is characterized by a specific energy, wavefunction (including spin), electron configuration, total angular momentum, and the way the orbital and spin angular momenta of the different electrons are coupled together. There are two descriptions for the coupling of angular momentum. One is called j-j coupling, and the other is called L-S coupling. The j-j coupling scheme is used for heavy elements (Z > 40), and the L-S coupling scheme is used for the lighter elements. L-S coupling also is called R-S or Russell-Saunders coupling.
L-S Coupling
In L-S coupling, the orbital and spin angular momenta of all the electrons are combined separately
\[L = \sum _i l_i \label{8.8.3}\]
\[S = \sum _i S_i \label {8.8.4}\]
The total angular momentum vector then is the sum of the total orbital angular momentum vector and the total spin angular momentum vector.
\[J = L + S \label {8.8.5}\]
The result of these vector sums is specified in a code that is called a Russell-Saunders term symbol, and each term symbol identifies an energy level of the atom. Consequently, the energy levels also are called terms. A term symbol has the form
\[\Large ^{2s+1} L_J\]
where the code letter that is used for the total orbital angular momentum quantum number L = 0, 1, 2, 3, 4, 5 is S, P, D, F, G, H, respectively. Note how this code matches that used for the atomic orbitals. The superscript \(2S+1\) gives the spin multiplicity of the state, where S is the total spin angular momentum quantum number. The spin multiplicity is the number of spin states associated with a given electronic state. In order not to confuse the code letter S for the orbital angular momentum with the spin quantum number S, you must examine the context in which it is used carefully. In the term symbol, the subscript \(J\) gives the total angular momentum quantum number. Because of spin-orbit coupling, only \(J\) and \(M_J\) are valid quantum numbers, but because the spin-orbit coupling is weak \(L\), \(M_L\), \(S\), and \(M_S\) still serve to identify and characterize the states for the lighter elements.
For example, the ground state, i.e. the lowest energy state, of the hydrogen atom corresponds to the electron configuration in which the electron occupies the 1s spatial orbital and can have either spin \(\alpha\) or spin \(\beta\). The term symbol for the ground state is \(^2 S_{1/2}\), which is read as “doublet S 1/2”. The spin quantum number is 1/2 so the superscript \(2S+1 = 2\), which gives the spin multiplicity of the state, i.e. the number of spin states equals 2 corresponding to \(\alpha\) and \(\beta\). The \(S\) in the term symbol indicates that the total orbital angular momentum quantum number is 0 (For the ground state of hydrogen, there is only one electron and it is in an s-orbital with \(l = 0\) ). The subscript ½ refers to the total angular momentum quantum number. The total angular momentum is the sum of the spin and orbital angular momenta for the electrons in an atom. In this case, the total angular momentum quantum number is just the spin angular momentum quantum number, ½, since the orbital angular momentum is zero. The ground state has a degeneracy of two because the total angular momentum can have a z-axis projection of \(+\frac {1}{2} \hbar\) or \(-\frac {1}{2} \hbar\), corresponding to \(m_J\) = +1/2 or -1/2 resulting from the two electron spin states \(\alpha\) and \(\beta\). We also can say, equivalently, that the ground state term or energy level is two-fold degenerate.
Exercise \(\PageIndex{1}\)
Write the term symbol for a state that has 0 for both the spin and orbital angular momentum quantum numbers.
Exercise \(\PageIndex{2}\)
Write the term symbol for a state that has 0 for the spin and 1 for the orbital angular momentum quantum numbers
Russell-Saunders Selection Rules
Higher energy or excited orbital configurations also exist. The hydrogen atom can absorb energy, and the electron can be promoted to a higher energy orbital. The electronic states that result from these excited orbital configurations also are characterized or labeled by term symbols. The details of how to determine the term symbols for multi-electron atoms and for cases where both the orbital and spin angular momentum differ from zero are given elsewhere, along with rules for determining the relative energies of the terms.
We have found that the selection rules for a single electron moving from one atomic orbital to another are
\[ \Delta l = \pm 1 \label{8.8.6}\]
\[ \Delta m_l = 0, \pm 1 \label {8.8.7}\]
For an atom as a whole in the limit of L-S coupling, the Russell-Saunders selection rules are
\[ \Delta S = 0 \label{8.8.8}\]
\[\Delta L = 0, \pm 1 \label{8.8.9}\]
\[\Delta J = 0, \pm 1 \label {8.8.10}\]
However, the \(J =0\) to \(J= 0\) transition is forbidden
\[\Delta m_J = 0, \pm 1 \label {8.8.11}\]
However, the \(m_J = 0\) to \(m_J = 0\) is forbidden if \(\Delta J = 0\)
These selection rules result from the general properties of angular momentum such as the conservation of angular momentum and commutation relations.
Now we want to apply these ideas to understand why multiplet structure is found in the luminescence spectrum of hydrogen and single electron ions. As we have said, the \(H_{\alpha}\) line in the Balmer series at 656.279 nm can be understood as due to a transition of an electron in a n = 3 atomic orbital to a n = 2 atomic orbital. When this spectral line was examined using high-resolution instruments, it was found actually to be a doublet, i.e. two lines separated by 0.326 cm-1.
There are 9 degenerate orbitals associated with the n = 3 level, and 4 associated with the n = 2 level. Since an electron can be in any orbital with any one of two spins, we expect the total number of states to be twice the number of orbitals. The number of orbitals is given by n2 so there should be 8 states associated with n = 2 and 18 states associated with n = 3. Using the ideas of vector addition of angular momentum, the terms that result from having an electron in any one of these orbitals are given in Table \(\PageIndex{1}\).
|
|
|
|
|---|---|---|
| 1s1 | 2S1/2 | 2 |
| 2s1 | 2S1/2 | 2 |
| 2p1 | 2P1/2, 2P3/2 | 2, 4 |
| 3s1 | 2S1/2 | 2 |
| 3p1 | 2P1/2, 2P3/2 | 2, 4 |
| 3d1 | 2D3/2, 2D5/2 | 4, 6 |
Table \(\PageIndex{1}\) shows that there are 3 terms associated with n = 2, and 5 terms associated with n = 3. In principle, each term can have a different energy. The degeneracy of each term is determined by the number of projections that the total angular momentum vector has on the z-axis. These projections depend on the mJ quantum number, which ranges from +J to – J in integer steps. J is the total angular momentum quantum number, which is given by the subscript in the term symbol. This relationship between mJ and J (mJ varies from +J to – J in integer steps) is true for any angular momentum vector.
Exercise \(\PageIndex{3}\)
Confirm that the term symbols in Table 8. are correct.
Exercise \(\PageIndex{4}\)
Confirm that the values for the degeneracy in Table \(\PageIndex{1}\) are correct and that the total number of states add up to 8 for n = 2 and 18 for n = 3.
The energies of the terms depend upon spin-orbit coupling and relativistic corrections that need to be included in the Hamiltonian operator in order to provide a more complete description of the hydrogen atom. As a consequence of these effects, all terms with the same n and J quantum numbers have the same energy while terms with different values for n or J have different energies. The theoretical term splittings as given by H.E. White, Introduction to Atomic Spectra (McGraw-Hill, New York, 1934) pp. 132-137. are shown in Figure \(\PageIndex{8}\).2.

Figure \(\PageIndex{2}\) shows 5 allowed transitions for the electron in the states associated with n = 3 to the states associated with n = 2. Of these five, two are most intense and are responsible for the doublet structure. These two transitions are indicated by the wide black lines at the bottom of the figure to correspond to the lines observed in the photographic spectrum shown in Figure \(\PageIndex{8}\).2. The other transitions contribute to the width of these lines or are not observed. The theoretical value for the doublet splitting is 0.328 cm-1, which is in excellent agreement with the measured value of 0.326 cm-1. The value of 0.328 cm-1 is obtained by taking the difference, 0.364 – 0.036 cm-1,in the term splittings.
As we have just seen, the electronic states, as identified by the term symbols, are essential in understanding the spectra and energy level structure of atoms, but it also is important to associate the term symbols and states with the orbital electron configurations. The orbital configurations help us understand many of the general or coarse features of spectra and are necessary to produce a physical picture of how the electron density changes because of a spectroscopic transition.
Exercise \(\PageIndex{5}\)
Use the Russell-Saunders selection rules to determine which transitions contribute to the \(H_{\alpha}\) line in the hydrogen spectrum.
Magnetic Field Effects
The Zeeman effect that was described in Section 8.4 only considered the orbital motion of the electron and did not include spin angular momentum and the spin magnetic moment. For a more complete analysis of the Zeeman effect associated with the n = 2 to n = 1 transition in the hydrogen atom, we need to use the term symbols for the states, examine how the mJ degeneracy is removed by the magnetic field, and determine which transitions between the states are allowed.
The states involved in a transition of an electron from the 2p atomic orbital to the 1s atomic orbital (where the hydrogen atom goes from the 2p1 configuration to the 1s1 configuration) are identified in Table \(\PageIndex{8}\).1. The 2p1 configuration produces 2P3/2 and 2P1/2 terms with the latter being lower in energy by 0.364 cm-1 as shown in Figure \(\PageIndex{8}\).2. The 1s1 configuration corresponds to a 2S1/2 term, which also is shown in Figure \(\PageIndex{2}\).
The orbital energy in a magnetic field was given by Equation \ref{8.8.12} , which is repeated here.
\[ \left \langle E \right \rangle = E_0 + \mu _B B_z m_l \label {8.8.12}\]
This equation can be generalized by changing the angular momentum quantum number to J and adding a g-factor to account for different gyromagnetic ratios
\[ \left \langle E \right \rangle = E_0 + gm_J \mu _B B_z \label {8.8.13}\]
While the g-factor equals 2 for a free electron or an electron in an s-orbital, the g-factor of an electron is affected by spin-orbit coupling. For the case of L-S coupling, the g-factor is given by
\[ g = 1 + \frac {J(J + 1) + S( S + 1) - L (L + 1)}{2J (J + 1)} \label {8.8.14}\]
To identify how the energy of states change in an electric field we need only consider the gmJ factor in Equation since B, which is the Bohr magneton, is a constant and the energy changes just scale with \(B_z\), which is the magnetic field. Consequently we can describe the splittings in terms of gmJ units where one gmJ unit is the product \(\mu _B B_z\).
Table \(\PageIndex{3}\) lists the quantities needed for the Zeeman effect analysis. The information in this table shows that the \(2P_{3/2}\) term splits into 4 components. Two components move up in energy 6/3 units and 2/3 units, respectively, and two move down –6/3 and –2/3 units. The 2P1/2 term splits into two components. One moves up 1/3 unit, and the other moves down 1/3 unit. The 2S1/2 also splits into two components, each moving 1 unit up and down, respectively. The energies of these states in a magnetic field along with the allowed transitions between them are illustrated in Figure \(\PageIndex{3}\). The addition of spin angular momentum has made the situation much more complicated. Previously we considered a Zeeman effect that produced 3 spectral lines from one, now 2 lines turn into 10 lines in a magnetic field. These 10 lines correspond to 10 different possible transitions of the electron from the 2p1 configuration to the 1s1 configuration. These transitions produce two multiplets in the spectrum, one of 6 lines and one of 4 lines.
|
|
|
|
|
|
|
|
|---|---|---|---|---|---|---|
|
\(2^P_{3/2}\)
|
3/2
|
1
|
1/2
|
4/3
|
3/2
|
6/3
|
|
1/2
|
2/3
|
|||||
|
-1/2
|
-2/3
|
|||||
|
-3/2
|
-6/3
|
|||||
|
\(^2P_{1/2}\)
|
1/2
|
1
|
1/2
|
2/3
|
1/2
|
1/3
|
|
-1/2
|
-1/3
|
|||||
|
\(^2S_{1/2}\)
|
1/2
|
0
|
½
|
2
|
1/2
|
1
|
|
-1/2
|
-1
|
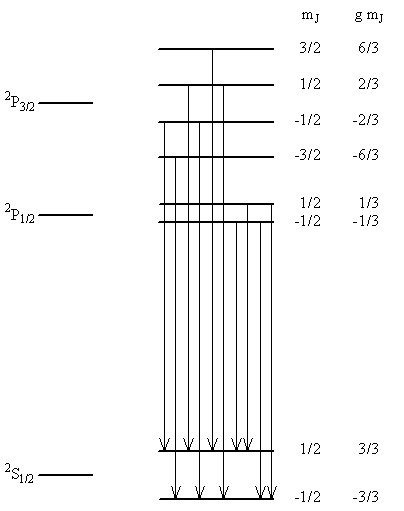
Exercise \(\PageIndex{6}\)
Using the information in Figure \(\PageIndex{8}\).3,(a) determine the spacing between the lines in the two multiplets in units of \(\mu _B B_z\),(b) determine the magnitude of \(\mu _B B_z\) </sub>for a field of 10,000 Gauss and for a field of 1 Tesla,(c) approximately what is the separation of the two multiplets in wavenumbers,(d) draw a sketch showing the field-on and field-off spectra you might record in the laboratory for the \(2p \rightarrow 1s\) transition, and(e) from the allowed transitions that are shown in the figure and considering the ones that do not occur, determine what the selections rules must be for ΔS, ΔL, ΔJ, and ΔmJ.


