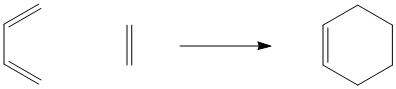PR3. The Diels Alder Reaction
- Page ID
- 4343
As noted previously, the Diels Alder reaction is a classic example of a pericyclic reaction.
Figure PR3.1. The Diels Alder reaction.
Unlike the Cope and Claisen rearrangements, this reaction often occur intermolecularly (between two molecules). It always occurs between an alkene on one molecule and a conjugated diene on the other molecule. The alkene is referred to as a "dienophile"; it reacts with the conjugated pair of double bonds.
Figure PR3.2. The diene and dienophile in the Diels Alder reaction.
Problem PR3.1.
Draw curved arrows to keep track of electrons in the Diels Alder reaction.
Problem PR3.2.
Draw the aromatic transition state of the Diels Alder reaction.
Problem PR3.3.
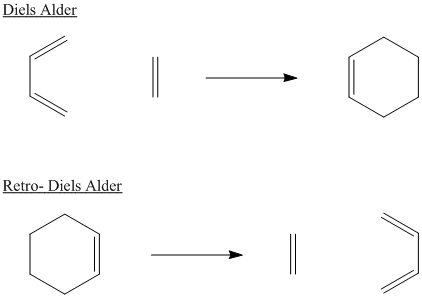
Many pericyclic reactions are reversible. The reversible process is usually named the same way as the forward reaction, but with the prefix "retro". For example, a retro-Diels Alder reaction is shown below.
- Draw curved arrows for the retro-Diels Alder reaction.
- The forward reaction is favoured at low temperature, whereas the retro reaction is favoured at high temperature. Explain why using the expression for free energy of a reaction, DG = DH - T DS.
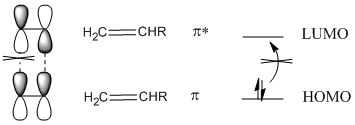
Once again, the reaction can be thought of in terms of a reorganization of electrons between these two molecules. In the Diels Alder reaction, we can think of an interaction between the LUMO on one molecule and the HOMO on the other. As it happens, the LUMO on one molecule has the correct symmetry such that it can overlap and form a bonding interaction with the HOMO on the other molecule.
Figure PR3.3. Qualitative molecular orbital picture of the Diels Alder reaction.
Pay attention to the p orbital drawings on the carbons that will bond to each other to form the six-membered ring. It is important that those orbitals are able to overlap with each other to form an in-phase interaction. In that way, these carbon atoms at the ends of the diene and dienophile are able to bond with each other.
A Diels Alder reaction is sometimes called a [2+4] addition reaction. A 2-carbon unit on one molecule interacts with a 4-carbon unit on another molecule.
In contrast, the addition of one regular alkene to another regular alkene would be called a [2+2] addition reaction. If this reaction occurred, two alkenes would come together to form a four-membered ring.
Figure PR3.4. A [2+2] addition reaction.
However, [2+2] addition reactions don't occur without special circumstances. There are a couple of reasons why, and you may be able to suggest some at this point.
You might say that the four-membered ring would be much more strained than the six-membered ring formed by the Diels Alder reaction. That is true, but it may not be reason enough to prevent the reaction from happening. Four-membered rings do occur in nature despite their strain energy.
You might also say that the benzene-like transition state that stabilizes the pathway through a Cope or Diels Alder reaction isn't possible in a [2+2] addition. In fact, the transition state would be more like antiaromatic cyclobutadiene. The transition state would be very high in energy.
Another problem shows up if we look at the orbital interactions in a [2+2] addition reaction. The HOMO on one alkene and LUMO on the other alkene do not overlap so that bonds can form between the two ends. If the p orbitals on one end are in phase, the p orbitals on the other end must be out of phase. The concerted reorganization of bonding possible for the Diels Alder reaction can't happen here.
Figure PR3.5. Qualitative molecular orbital picture of [2+2] addition reaction.
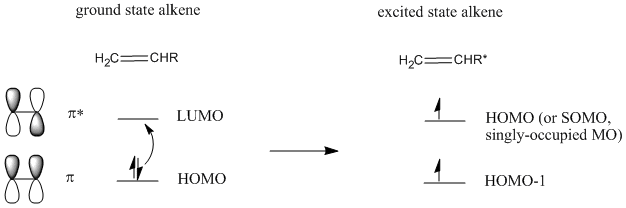
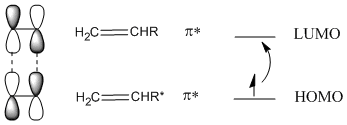
In fact, there is a way around that problem. Irradiating an alkene with UV light leads to promotion of an electron from the LUMO to the HOMO. The alkene is now in an "excited state".
Figure PR3.6. Excitation of an electron in an alkene.
This does not happen with 100% efficiency, so only some of the alkenes will become excited. In the excited state alkene, the HOMO now resembles the LUMO of the ground state alkene. Because of the matching symmetry between these orbitals, the addition reaction can proceed.
Figure PR3.7. HOMO-LUMO interaction between a ground-state alkene and an excited-state alkene.
A [4+2] reaction is sometimes referred to as "thermally-allowed", whereas a [2+2] addition is sometimes referred to as "photolytically-allowed." This distinction refers to the need for electronic excitation to accomplish the latter type of reaction.