CO9. Enolate Addition and Homologation
- Page ID
- 4239
CO9. Enolate Addition
Enolate ions are just another example of anionic nucelophiles. The reason they get a page to themselves is that they are especially important, especially in biological chemistry. They are also important in the synthesis of organic compounds, such as in the pharmaceutical industry.
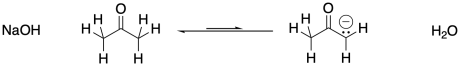
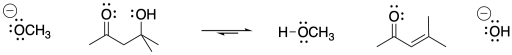
An enolate ion is the anion that forms when a proton is removed next to a carbonyl. The carbon next to the carbonyl is called the α-position (alpha position). The alpha position is acidic both because of the amount of positive charge on a proton in that position and because of the stability of the anion that results if that proton is removed.
Problem CO9.1.
Show why an enolate ion, such as the one formed from 2-propanone, above, is particularly stable.
Problem CO9.2.
Show a mechanism, with curved arrows, for the formation of the enolate ion from 2-propanone, above.
In the example above, 2-propanone is deprotonated at the α position to form the corresponding enolate ion. Note that sodium hydroxide is not a strong enough base to convert all of the 2-propanone to its enolate. The resulting enolate is basic enough to pull a proton from a water molecule, so an equilibrium results.
Negative charges are fairly stable on oxygen atoms. That allows this particular reaction to shift back to the left again. To make the reaction go all the way to the right, we would need a less stable anion on the left. That would make that anion more basic. Can you think of atoms that would be less stable as anions than oxygen?
The most commonly used very strong bases in synthetic chemistry involve anions of carbon, nitrogen or hydrogen. Some examples of compounds used as very strong bases are sodium hydride (NaH), sodium amide (NaNH2), lithium diisopropylamide (LiN[CH(CH3)2]2), and butyllithium (CH3CH2CH2CH2Li).
- Enolate ions form in equilibrium with their paent carbonyl compounds if a moderately strong base like sodium hydroxide is used.
- A very strong base, like sodium amide (NaNH2) or sodium hydride (NaH), would result in complete enolate formation.
However, it is sometimes really useful to have an equilibrium between a carbonyl compound and its enolate. That situation allows both a ketone (the 2-propanone, left) and its enolate (right) to be present at the same time. That means there is both a nucleophile and an electrophile (the ketone and the enolate). They will be able to react together.
- Simple carbonyls are electrophiles.
- The enolate ions that form from simple carbonyls are nucleophiles.
- Carbonyls react with enolate ions.
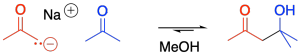
The reaction of an enolate nucleophile with another carbonyl compound is called an aldol reaction. A simple example of this reaction is shown here. This example involves the reaction of 2-propanone with its enolate.
Problem CO9.3.
Provide a mechanism, with curved arrows, for the aldol reaction of 2-propanone, above.
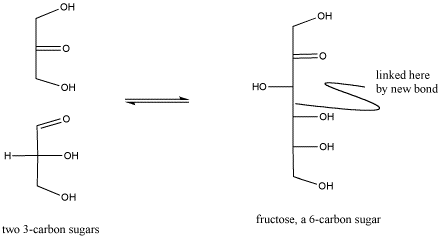
The biosynthesis of sugars, such as fructose, involves coupling smaller sugars together. If one sugar is converted into a nucleophile, it can donate electrons to the carbonyl on the other sugar, forming a new C-C bond. The carbonyl on the second sugar becomes a hydroxyl group in the new, larger sugar.
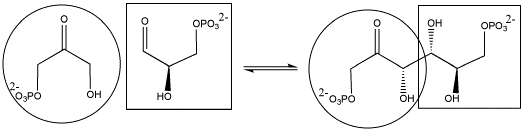
In the cell, sugars are typically in a phosphorylated form when they react in this way. Phosphorylation is often an important step in activating molecules for biochemical reactions.
Problem CO9.4.
Show the mechanism for the formation of the phosphorylated fructose shown above.
Sometimes, aldol reactions are followed by a subsequent reaction, called an elimination reaction. That reaction formally produces a molecule of water. Early studies of this reaction would result in droplets of condensation on the glassware in which the reaction occured; hence, it is sometimes called a condensation reaction.
Problem CO9.5.
Provide a mechanism for the aldol condensation shown above.
It can be hard to predict the outcome of an aldol reaction because of the fact that there are two possible products from an aldol reaction (one with a new hydroxyl and one with a new double bond). A chemist might try to make one product in the laboratory, and end up with the other. This process can be difficult to control. However, in general, the elimination reaction is encouraged by heating the reaction. The reaction sometimes occurs without elimination if the reaction is kept cool. However, there are also other factors that may come into play.
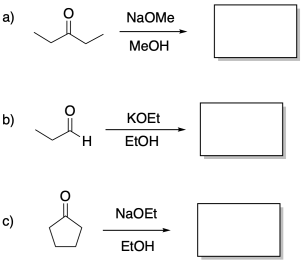
Problem CO9.7.
The following compound would give multiple products through different aldol condensation reactions. Show the products.
Problem CO9.8.
Sometimes, two different compounds may react together in an aldol reaction. The following compounds would give multiple products through different aldol condensation reactions. Show the products.
Problem CO9.9.
Only some of the following compounds may undergo aldol reactions. Select which ones may not undergo the reaction, and explain what factor prevents them from reacting.
Problem CO9.10.
Fill in the products of the following aldol condensations.
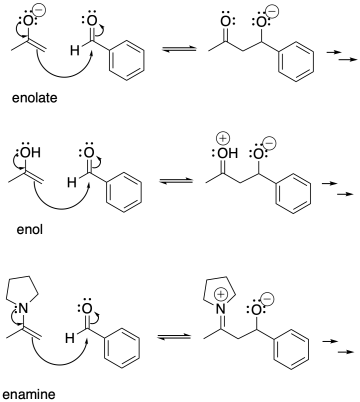
Aldol reactions do not just occur with enolate anions, however. Enols are the neutral form of enolates, protonated on the oxygen instead of the alpha carbon. Enols are also good nucleophiles. In an enol nucleophile, the pi bond acts as the electron source, rather than the lone pair. However, the pi bond gets a boost from the lone pair on the oxygen.
Enols are always present in equilibrium with aldehydes and ketones. An enol is a simple tautomer of a carbonyl compound. To get from one to the other, a proton is simply transferred from one position to the other.
Enamines are also good nucleophiles for aldol-type reactions.
Problem CO9.11.
Show the subsequent protonation step in the reactions involving the enol and the enamine above.
Problem CO9.12.
An enamine reaction is usually followed by hydrolysis of the C=N bond in the iminium ion. Show the mechanism for conversion of the iminium ion to the carbonyl.