7.3: Inert Atmospheric Methods
- Page ID
- 93201
 |
 |
 |
 |
|
Prepare the balloon Fill a balloon with an inert gas (nitrogen or argon) to 7-8 inches in diameter. Twist the balloon to prevent gas from escaping, then attach a needle and insert into a rubber stopper to plug. |
Prepare the reaction flask Flame or oven dry a reaction flask (with stir bar), and fold a rubber septum over the joint while wearing thick gloves. Clamp the hot flask to the ring stand or latticework and insert the balloon of inert gas. Insert an "exit needle" and allow the flask to flush for ~5 minutes (to displace the air). Remove the exit needle and allow the flask to cool. |
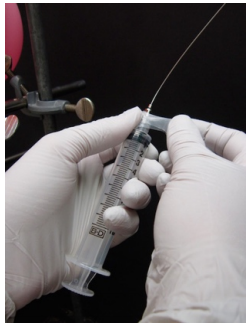
Prepare the syringe Obtain a needle from the hot oven and screw it into the tip of a freshly opened plastic syringe. Wrap the needle/syringe joint with Teflon tape or Parafilm. Flush the syringe with inert gas by using an empty flask attached to a balloon of inert gas. Withdraw a volume of gas and expunge into the air. Insert the needle tip into a rubber stopper to plug. |
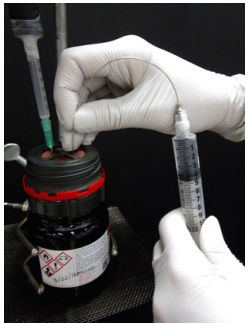
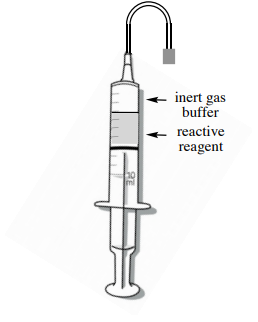
Withdraw the reagent Use the prepared syringe to slowly withdraw some reagent (or air may enter the syringe). Push out the gas bubble. Withdraw reagent to a slightly greater volume than needed, then expunge liquid to the exact volume. Place the needle in the headspace of the bottle and withdraw an "inert gas buffer", \(\sim 20\%\) of the volume of the syringe. Insert the needle into a rubber stopper for transport. |
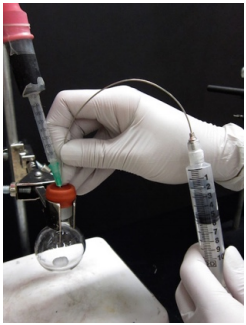 |
Deliver the reagent Into the reaction flask (with inert gas balloon), deliver first the inert gas buffer, then the air-sensitive reagent. Don't push out the residual liquid. |
 |
Withdraw an inert gas buffer into the mostly empty syringe. Clean by rinsing with two portions (few \(\text{mL}\) each) of solvent (similar to reagent solvent), then one portion of water and two portions of acetone. |


