7.13: Single Solvent Crystallization
- Page ID
- 93211
 |
 |
 |
 |
|
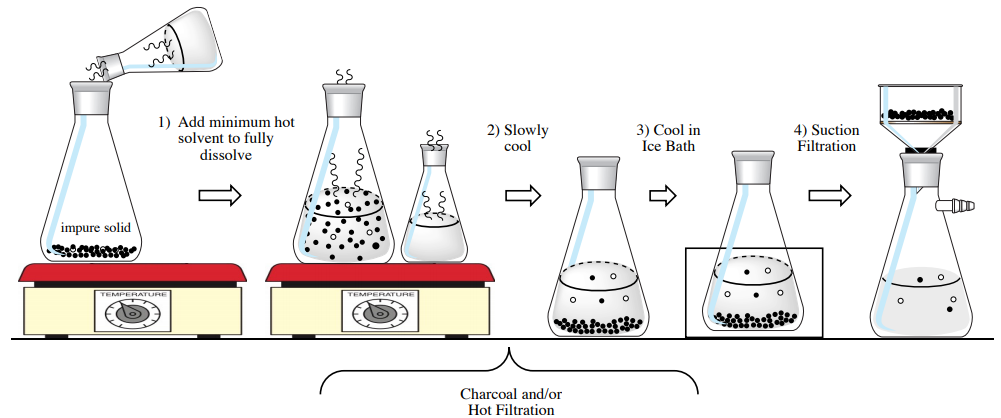
Heat some solvent on a heat source to a boil (include boiling stones). To the impure solid in an Erlenmeyer flask, add a small portion of hot solvent. If a hot filtration step is expected, use boiling stones with the impure solid, or a boiling stick or stir bar if a filtration is not expected. Put both flasks on the heat source and bring to a boil. |
Add the minimum amount of boiling solvent needed to dissolve the impure solid, with swirling: For \(100 \: \text{mg}\)-\(1 \: \text{g}\) quantities,add \(0.5\)-\(2 \: \text{mL}\) solvent each time. For smaller scales, add solvent dropwise. Wait for each portion of solvent to come to a boil before adding more. Possibly use charcoal and hot filtration at this point if colored or insoluble impurities are present. |
When dissolved, remove the solution from the heat source and remove the boiling stick or stir bar if used. Allow the solution to slowly cool atop some paper towels,and with a small watch glass over the mouth of the Erlenmeyer. If a hint of cloudiness is seen in the solution, or if the solution has cooled a good deal without crystallizing, scratch with a glass stirring rod to initiate crystallization. |
A proper crystallization takes between 5-20 minutes to complete. When the solution is at room temperature, place it in an ice bath for at least 10 minutes to maximize crystal formation. Also chill a rinse solvent in the ice bath. Collect the crystals by suction filtration. |


