5.2A: Uses of Simple Distillation
- Page ID
- 95717
Concentration of Alcohol
Fermented grains can produce a maximum alcohol content of about \(16\%\), the upper limit of most beers and wines, as the yeast organism used in fermentation cannot survive in more concentrated alcoholic solutions. However, spirits such as gin, vodka, and rum usually have an alcohol content of roughly \(40\%\) by volume. Distillation is the method used to concentrate fermented solutions and produce hard liquors. A distillation of fermented grape juice is commonly done in academic laboratories (Figure 5.2).

Figure 5.3 shows the distillation of red wine. Notice how the wine is noticeably purified in the process as the distillate is clear (Figure 5.3d). All concentrated alcoholic products at first distill clear, but some become colored during the aging process (from components leaching out of the barrels they are stored in, or from oxidation).

The distillation of red wine can be proven to have concentrated the alcohol through a flame test, which is the origin of the term "proof". In the 16\(^\text{th}\) century when rum was a bartering item, it was regularly tested to ensure that it had not been watered down. An alcoholic sample that is \(57\%\) alcohol by volume is capable of catching on fire, while more dilute solutions cannot, enabling ignition to serve as "proof" of alcohol content. The red wine used in Figure 5.3 did not ignite, which is consistent with its label that states it to be only \(13\%\) alcohol (Figure 5.4a). The distillate easily caught on fire (Figure 5.4c+d), and density analysis showed it to be \(67\%\) alcohol.

Distilled Water
Access to clean, fresh water is a major problem facing the world today. In countries neighboring the ocean, seawater desalination is sometimes used to provide the country with drinkable water. Distillation is one of the main methods\(^1\) used to purify ocean water and works well since salt, microorganisms, and other components of seawater are non-volatile. The main disadvantage of distilling water is that the process requires a lot of energy, and unless engineered creatively, the economics can be a major deterrent to using the method. Heat released from a power plant is often used to provide the energy for the distillation of seawater, and Saudi Arabia and Israel mainly use coupled power plants and distilleries to obtain roughly half the fresh water needed for their countries.\(^2\)
Along with drinking water, distilled water is necessary for scientific lab work as dissolved salts in tap water may interfere with some experiments. Many science buildings have their own distilled water generators (Figure 5.5a), enabling wash bottles and carboys to be filled with a turn of the spigot (Figure 5.5b).

Purification of Reagents and Products
Distillation is an excellent purification tool for many liquids, and can be used to purify products from a chemical reaction. Figure 5.6 shows the distillation of a crude sample of isoamyl acetate, formed through a Fischer esterification reaction. The crude sample was originally yellow (Figure 5.6a), but the distillate was colorless (Figure 5.6d), making obvious the removal of some materials through the process.

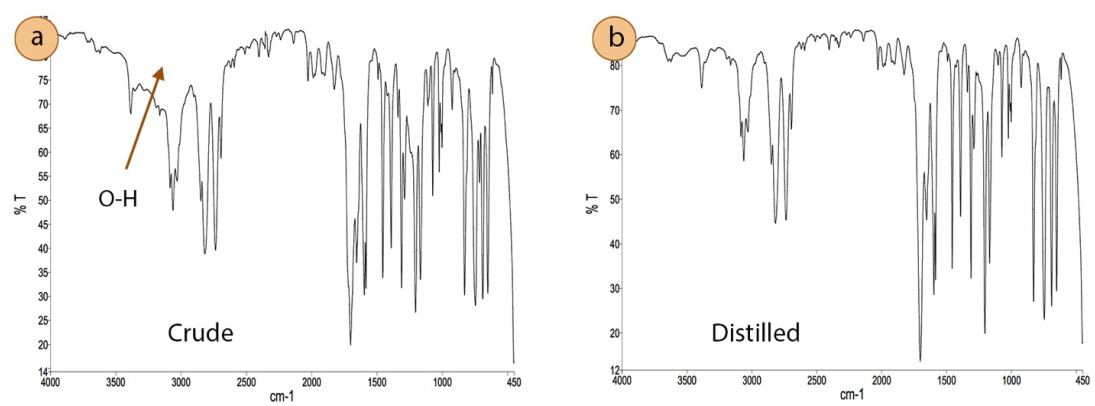
Distillation can also be used to purify reagents that have degraded over time. Figure 5.7a shows a bottle of benzaldehyde that has partially oxidized, as evidenced by the crystals of benzoic acid seen adhering to the inside of the glass (indicated with an arrow). A simple distillation purified the benzaldehyde sample, and its effectiveness was demonstrated through comparison of the infrared (IR) spectra of the original material (Figure 5.8a) and the distillate (Figure 5.8b). The broad region indicated by an arrow in Figure 5.8a represent the \(\ce{O-H}\) stretch of a carboxylic acid (benzoic acid), and the distillate's IR spectrum lacks this feature.

\(^1\)More often, vacuum distillation is used for desalination, as the lowered pressure allows for boiling to occur at a lower temperature (which requires less energy). Vacuum distillation of seawater is still energy intensive. Reverse osmosis is also used for seawater desalination.
\(^2\)Pyper, Julia, "Israel is creating a water surplus using desalination," E+E News, February 7, 2014.


