17.1: Some Mechanistic Concepts in Electrophilic Addition Reactions to C=C Bonds
- Page ID
- 216685
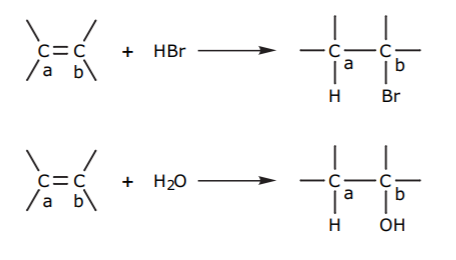
The C=C bond is considered to be a weak base/nucleophile. The high concentration of electron density makes the pi bond a Lewis base, but in order to donate electrons the pi bond must be broken first. In a reaction which is basically the reverse of elimination, an electrophile of general formula E-Y can add to the C=C double bond. E represents the electrophilic center, and Y is the rest of the molecule, or nucleophilic part. Proton acids such as HCl fit this description, with E = H and Y = Cl. Water can also be viewed as an electrophile with E = H and Y = OH. In a generalized representation, the electrophile E-Y adds to the pi bond, converting the sp2 carbons into sp3 carbons.
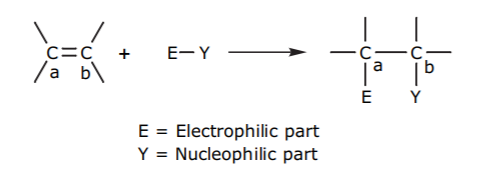
Examples: Addition of HBr and water to the π-bond