14.6: Sn1 Reactions and Rearrangements Involving Primary Substrates
- Page ID
- 216040
Primary substrates normally do not follow Sn1 mechanisms because they do not form stable cations. However, a hindered primary substrate can be forced into an Sn1 mechanism if sufficient energy and time are allowed, for example boiling the substrate in a nucleophilic solvent such as ethanol.
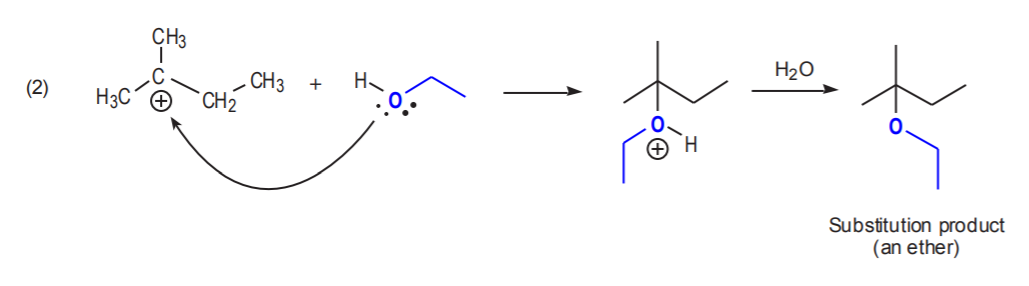
The nucleophile cannot do a backside attack, and the substrate cannot form stable cations. In this case the substrate will begin to rearrange as the leaving group departs. This avoids formation of a primary cation. As the leaving group departs, a positive charge begins to develop on the carbon bearing the leaving group, and the rearrangement process starts, all in unison. The reaction of neopentyl bromide with ethanol illustrates this point.

As the positive charge develops on the primary carbon while bromine leaves, the methyl group is migrating to an adjacent position to forma more stable cation.