11.5: Hydrogen Atoms and Protons in Organic Molecules
- Page ID
- 215760
A hydrogen atom that has lost its only electron is sometimes referred to as a proton. That is because once the electron is lost, all that remains is the nucleus, which in the case of hydrogen consists of only one proton.
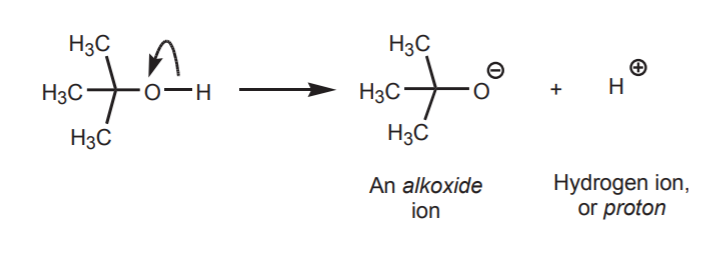
The large majority of organic reactions, or transformations, involve breaking old bonds and forming new ones. If a covalent bond is broken heterolytically, the products are ions. In the following example, the bond between carbon and oxygen in the t-butyl alcohol molecule breaks to yield a carbocation and hydroxide ion.
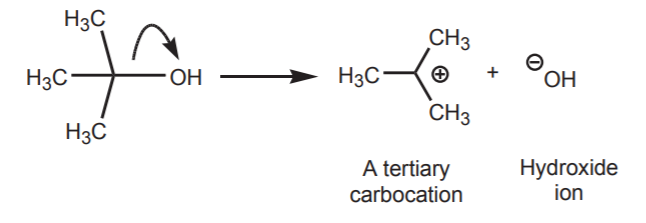
The full-headed curved arrow is being used to indicate the movement of an electron pair. In this case, the two electrons that make up the carbon-oxygen bond move towards the oxygen. The bond breaks, leaving the carbon with a positive charge, and the oxygen with a negative charge. In the absence of other factors, it is the difference in electronegativity between the two atoms that drives the direction of electron movement. When pushing arrows, remember that electrons move towards electronegative atoms, or towards areas of electron deficiency (positive, or partial positive charges). The electron pair moves towards the oxygen because it is the more electronegative of the two atoms.
If we examine the outcome of heterolytic bond cleavage between oxygen and hydrogen, we see that, once again, oxygen takes the two electrons because it is the more electronegative atom. Hydrogen is left with only a positive charge. In other words, it becomes a proton.