10.2: Energy Profiles and Reaction Profiles
- Page ID
- 215746
A reaction profile is a plot of free energy (y-axis) vs. reaction progress (x-axis, or time axis). It is basically a movie played frame by frame. The x-axis represents sequential time events, or stages of the reaction. The y-axis represents the free energy associated with the structural changes taking place during the transformation.
As an example, consider the potential energy diagram of n-butane for rotation around the C2-C3 axis. For each stage during the rotation sequence (x-axis) there is a potential energy level associated with the particular structure that results from the rotation.
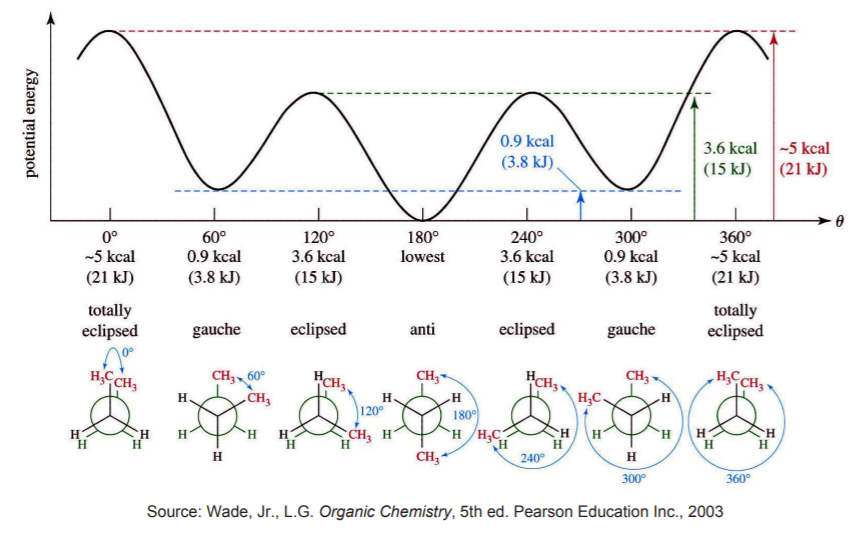
As an analogy (and an excuse to have a little fun), play a car chase sequence from an action movie frame by frame. The “low energy” images (e.g. when the chase is just beginning) have more definition than the “high energy” images (at the height of the chase), which appear more blurred.


