4.2: Common Bonding Patterns for 1st and 2nd Row Elements
- Page ID
- 214225
Once we write enough Lewis formulas containing the elements of interest in organic chemistry, which are mostly the second row elements, we find that certain bonding patterns occur over and over. Learning these patterns is useful when trying to write Lewis formulas because they provide a convenient starting point. For example, in several of the structures given in the previous section, we find that the carbon bonded to three hydrogens is a unit that occurs quite frequently. It is called the methyl group, represented by CH3. It is so common that it is valid to write it as such in Lewis formulas, even though it is in fact an abbreviated form, because everybody knows what it stands for.
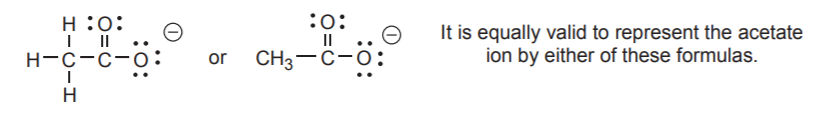
Other common bonding patterns are shown below.
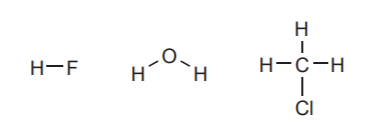
HYDROGEN:
Usually forms only one bond.
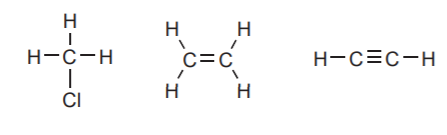
CARBON:
Forms four bonds when neutral, but it can also have only three bonds by bearing a positive or a negative charge. When it bears a negative charge it should also carry a pair of unshared electrons.
 Neutral carbon
Neutral carbon
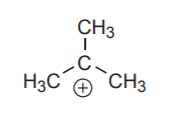 A carbocation has a central carbon with an incomplete octet and a formal +1 charge.
A carbocation has a central carbon with an incomplete octet and a formal +1 charge.
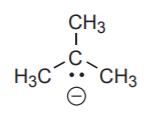 A carbanion has a central carbon with an unshared electron pair and a formal -1 charge.
A carbanion has a central carbon with an unshared electron pair and a formal -1 charge.
NITROGEN:
Forms three bonds and carries a lone pair of electrons when neutral. It can also form four bonds by bearing a positive charge, in which case it carries no unshared electrons. Finally, it can also form two bonds as it carries two unshared electron pairs and a negative charge.
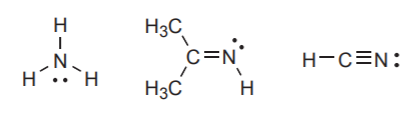 Neutral nitrogen
Neutral nitrogen
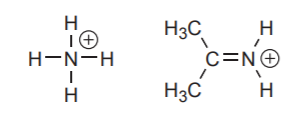 Positively charged nitrogen
Positively charged nitrogen
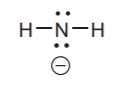 Negatively charged nitrogen
Negatively charged nitrogen
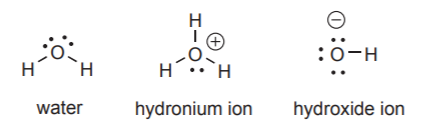
OXYGEN:
Forms two bonds and carries two lone pairs when neutral. It can form three bonds with a positive charge, or one bond with a negative charge. In each case it must carry the appropriate number of unshared electron pairs to complete the octet.
HALOGENS:
Form one bond and carry three electron pairs when neutral. Can carry a negative charge with no bonds. They are rarely seen with positive charges.
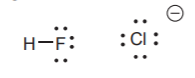
THIRD ROW ELEMENTS:
They behave like their second-row counterparts, except that they can expand their valence shells if needed.
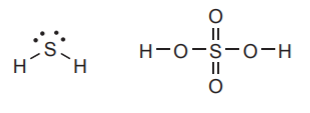 Electron pairs on oxygen are not shown for clarity.
Electron pairs on oxygen are not shown for clarity.

ELECTRON DEFICIENCY IN SECOND ROW ELEMENTS:
One thing worth noting is that, in the second row, only boron and carbon can form relatively stable species in which they bond with an incomplete octet. Examples have already been discussed. Boron has no choice but to be electron deficient. Carbon can bond with a complete octet or with an incomplete octet. Obviously bonding with a complete octet provides higher stability.
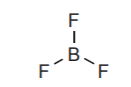 Boron has no choice but to have an incomplete octet
Boron has no choice but to have an incomplete octet
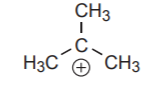 An electron deficient carbon in a carbocation
An electron deficient carbon in a carbocation
It is however very rare to observe species where nitrogen or oxygen bond with incomplete octets. Their high electronegativity renders such situation high energy, and therefore very unstable. For all intents and purposes, avoid writing formulas where oxygen or nitrogen are shown with incomplete octets, even if they carry a positive charge.
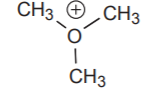 To write this structure without the lone pair of electrons on oxygen is unacceptable.
To write this structure without the lone pair of electrons on oxygen is unacceptable.
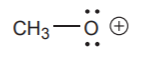 This structure is unacceptable and indeed it looks quite awkward
This structure is unacceptable and indeed it looks quite awkward
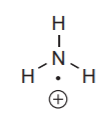 This species might exist in the high energy environment of a mass spectrometer, but it is not frequently observed in common organic reactions.
This species might exist in the high energy environment of a mass spectrometer, but it is not frequently observed in common organic reactions.


