21.2: Nucleophilic Acyl Substitution Reactions
- Page ID
- 36399
After completing this section, you should be able to
- compare the reactions of carboxylic acid derivatives with nucleophiles to the reactions of aldehydes and ketones with nucleophiles.
- arrange a given list of carboxylic acid derivatives in order of increasing or decreasing reactivity towards nucleophiles.
- explain the difference in reactivity towards nucleophiles of two or more given carboxylic acid derivatives.
- explain why esters and amides are commonly found in nature, but acid halides and acid anhydrides are not.
The general nucleophilic acyl substitution reaction, and its mechanism, were discussed earlier in “III. General Reactions of Carbonyl Compounds.” Review if necessary.
The reading describes the relative reactivities of “biologically relevant acyl groups.” Both acid anhydrides and acid halides readily react with water and cannot exist for any length of time in living organisms. The following scheme illustrates the relative reactivities of most carboxylic acid derivatives that you will encounter.
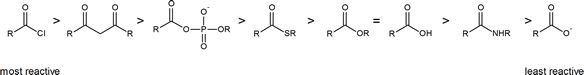
Carboxylic acid derivatives and acyl groups
Carboxylic acid derivatives can be distinguished from aldehydes and ketones by the presence of a group containing an electronegative heteroatom - usually oxygen, nitrogen, sulfur or phosphorus – bonded directly to the carbonyl carbon and represented by the symbol Y. The rest of the carboxylic acid derivative is called the acyl group which is made up of the carbonyl group and the attached alkyl group (R). Being electronegative, the Y group has the potential of receiving electrons from the alkoxide intermediate created during a nucleophilic acyl substitution and acting as a leaving group. Elimination of a leaving group allows for carbonyl bond reformation and C-Y bond cleavage to complete a substitution reaction.
The stability of a negative charge on a Y group can be gauged by the pKa of its corresponding conjugate acid (HY). A low conjugate acid pKa implies that the Y leaving group is a weak base with a stable negative charge and thus would make an efficient leaving group. Likewise, a high conjugate acid pKa means the Y group would likely make a poor leaving group. As shown in the table below, the conjugate acids of the possible carbide (R:-) and hydride (H:-) leaving groups of aldehydes and ketones have a relatively high pKa, while those of the Y leaving groups of carboxylic acid derivatives are much lower. In fact, the stability of the leaving groups is an important factor in determining the reactivity of different carboxylic acid derivatives toward nucleophilic acyl substitutions. Based on the pKa data we can deduce that amides will most likely be the least reactive of the carboxylic acid derivatives.
Carbonyl Compounds and their Leaving Groups
| Name | Carbonyl Compound | Leaving Group | Conjugate Acid of the Leaving Group | pKa |
| Acid Chloride | Cl- | H-Cl | -7 | |
| Acid Anhydride | 3-5 | |||
| Ester | -OR' | H-OR' | 15-16 | |
| Carboxylic Acid | -OH | H-OH | 15.7 | |
| Amide | -NH2 | H-NH2 | 36 | |
| Ketone | -R' | H-R' | 50 | |
| Aldehyde | -H | H-H | Very Large |
What is the ‘Y’ group in:
- an acid anhydride?
- a carboxylic acid?
Answers
a.
b.
The Mechanism of Nucleophilic Acyl Substitution
Aldehydes and ketones, along with carboxylic acid derivatives all have the C=O carbonyl bond in common. Thus, the electrophilic character of the carbonyl carbon plays an important part in the reactivity of all of these compounds. Because the carbonyl carbon of aldehydes and ketones do not contain suitable leaving groups, their primary reaction is fundamentally different than carboxylic acid derivatives. Remember that aldehydes and ketones tend to undergo nucleophilic addition to form a tetrahedral alkoxide intermediate. Once formed, the negative charge of the alkoxide intermediate cannot be transferred to other substituents because the potential carbide (R:-) and hydride (H:-) leaving groups are too unstable. Instead the negative charge is held by the alkoxide until protonation converts it into an alcohol.
Carboxylic acid derivatives tend to undergo a reaction called nucleophilic acyl substitution. In the same fashion as nucleophilic addition, this mechanism starts with a nucleophilic attack on an electrophilic carbonyl carbon, forming a tetrahedral alkoxide intermediate. The alkoxide negative charge can gain stability by being transferred to the Y leaving group. Elimination of the Y leaving group in the second mechanistic step, allows the C=O carbonyl bond to reform thus creating a new acyl compound. The reaction is considered a substitution due to the Y group of the carboxylic acid derivative being exchanged with an incoming nucleophile.
1) Nucleophilic Addition
2) Leaving Group Removal
The Relative Reactivity of Carboxylic Acid Derivatives
The relative reactivity of carboxylic acid derivatives is an important concept for entering into a detailed examination of nucleophilic acyl substitutions. There are two main concepts directly relating to the mechanism which when combined lead to an overall explanation of the differences in reactivity: the stability of the carbonyl and the effectiveness of the Y leaving group.
Carbonyl Stability
The rate of the first mechanistic step is mainly affected by the stability of the carbonyl moiety. Stabilization reduces the electrophilic character of the carbonyl carbon by reducing its partial positive charge which in turn reduces the rate of this step of the mechanism. The ability of substituents attached to the carbonyl carbon to donate or withdraw electron density is the primary factor determining carbonyl stabilization. As previously discussed, the presence of two electron donating alkyl substituents reduces the partial positive charge on the carbonyl carbon of ketones making them less reactive to nucleophilic attack than aldehydes. The Y group heteroatom's ability to stabilize a carbonyl by donating electrons through resonance makes most carboxylic acid derivatives even less reactive. Overall, delocalization of the carbonyl carbon's partial positive charge onto the adjacent Y group heteroatom reduces the electrophilic character of the carbonyl. The electronegativity of the Y group heteroatom generally determines extent of this stabilization. The less electronegative the Y group heteroatom the better it is able to stabilize the resonance structure with the delocalized positive charge reducing the carbonyl's electrophilicity.
Leaving Group Ability
The leaving group ability of the Y group is the most important factor in determining the rate of the second mechanistic step of nucleophilic acyl substitution. As discussed above, the effectiveness of a leaving group is related to its ability to stabilize a negative charge particularly through having a relatively high electronegativity or having the ability to delocalize the negative charge through resonance. The Y group structural features which allow for the stabilization of a negative charge also allow for the stabilization of the transition state of the second step of the mechanism. Overall, the better the leaving group ability of the Y group, the higher the rate of second step of the mechanism.
These two effects, carbonyl stability and leaving group ability, when combined predict the relative reactivity of carboxylic acid derivatives. Fortunately, the effects tend to work synergistically. Y groups that are effective leaving groups also tend to poorly stabilize carbonyls through resonance making reaction rates of nucleophilic acyl substitution higher. Likewise, poor leaving groups tend to effectively stabilize carbonyls through resonance and reduce reaction rates. In fact, depending on the carbonyl structure, either step one or step two of the mechanism can be the rate determining step. For Y groups with a poor leaving group ability, the second step of the mechanism is rate determining because of the weakly stabilized transition state. The first step of the mechanism is rate determining for Y groups with good leaving group ability because they tend to stabilize the starting material's carbonyl increasing the energy distance to the first transition state.
How the two effects actually combine to produce the overall reactivity for each carboxylic acid derivative is slightly different. As comparison of one of the more reactive carboxylic acid derivatives, acid chlorides, and one of the least reactive, amides, will be used as a discussion.
In amides, the nitrogen atom is a powerful electron donating group by resonance. Nitrogen is less electronegative than oxygen and can therefore better stabilize the resonance structure of the delocalized positive charge than most other acid derivatives. This lowers the energy of the starting material which increases the overall energy barrier. Likewise, -NH2 is a relatively poor leaving group (since it is the conjugate base of a very weak acid RNH2) which increases the transition state energy of the second step of the mechanism also increasing the overall energy barrier. The two effects combine to increase the overall energy barrier which must be overcome during the reaction, causing reaction rates to decrease and making amides very unreactive toward nucleophilic acyl substitution.
In acid chlorides the -Cl is a very effective leaving group making the energy barrier of the second step relatively small. However, this does not have a significant effect since the first step of the mechanism is rate determining. The resonance stabilization offered by the chlorine atom is ineffective due to the poor overlap between the chlorine 3p orbital and the carbon 2p orbital. The poor overlap means that acid chlorides are much less stabilized by resonance than amides. In addition, since chlorine is more electronegative than nitrogen was in the amide example, the inductive electron-withdrawing effects of chlorine become important. This destabilizes the carbonyl and increases the electrophilic character of the carbonyl carbon. The energy barrier of the first step of the mechanism is small due to the lack of stabilization in the acid chloride starting material. Therefore the overall rate of reaction for acid chlorides is high making them very reactive towards nucleophilic acyl substitutions.
As a general rule, stabilizing effects from the Y heteroatom tends to make carboxylic acid derivatives less reactive toward the initial nucleophilic attack than aldehydes and ketone. One notable exception is acid chlorides, where the destabilizing inductive electron-withdrawing properties of the chlorine outweigh the resonance stabilizing effects making them more reactive towards nucleophilic attack. This point will become important later in the chapter where it will be used to explain the outcome of certain reactions.
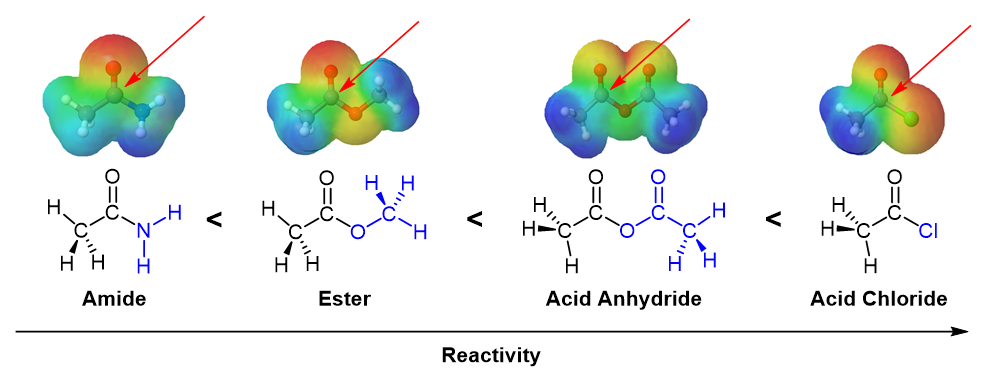
The reactivity of a carboxylic acid derivative can be visualized by using an electrostatic potential map to look at the electron density around the carbonyl carbon. A blue/green color represents the amount of positive charge present. The carbonyl carbons of the amide and ester have a relatively small amount of positive charge making them among the least reactive toward nucleophilic attack. The acid chloride's and anhydride's carbonyl carbon has more of a blue/color color indicating a higher positive charge and making them more reactive than the amide and ester.
Acid Derivative Interconversion
From this understanding of reactivity, multiple step synthesis strategies can be developed. Typically more reactive carboxylic acid derivatives can be used to synthesize less reactive derivatives as illustrated in the diagram below. As will be shown later in this chapter, highly reactive acid chlorides, when combined with the appropriate nucleophile, can be directly converting into acid anhydrides, thioester, esters, and amides. However, the less reactive amides cannot be directly converted into ester, thioester, anhydrides, or acid chlorides. It is possible to make these conversions with an amide but multiple reaction steps are required.
The nucleophiles required to make the conversion from one carboxylic acid derivative to another are common and have been discussed multiple times in this text. Because the same nucleophile is typically used to convert to a specific carboxylic acid derivative conversion, the reactions are given generic names.
- Hydrolysis: A reaction with water as a nucleophile to create a carboxylic acid.
- Alcoholysis: A reaction with an alcohol nucleophile to create an ester.
- Aminolysis: A reaction with ammonia or an amine nucleophile to create an amide.
- Reduction: A reaction with a hydride nucleophile to create an aldehyde or 1o alcohol.
- Organometallic: A reaction with a carbanion nucleophile from an organometallic reagent to create a ketone or a 3o alcohol.
Predicting the Product of a Nucleophilic Acyl Substitution Reaction
There are two major pieces of a nucleophilic acyl substitution reaction which need to be identified in order to predict the product: the leaving group of the carboxylic acid derivative (Y) and the nucleophile. It is important to identify if the nucleophile is neutral (i.e. water, alcohols, ammonia, amines) or negatively charged (i.e. hydroxide, alkoxides, hydrides, carbanions). Also, it is important to note that certain hydride and organometallic reagents are capable of a double addition to carboxylic acid derivatives where the first step is a nucleophilic acyl substitution and the second is a nucleophilic addition.
Negatively Charged Nucleophiles
Neutral Nucleophiles
Worked Example
Draw the expected products of the following reactions:
a)
b)
Solutions
a) The nucleophile is neutral.
b) The nucleophile is negatively charged.
Examples
1) Which of the following compounds would be expected to be the most reactive towards nucleophilic acyl substitution? Briefly explain your answer.
a)
b)
2) Draw the expected products of the following reactions:
a)
b)
c)
Solutions
1)
a) Compound A would expected to be the more reactive. For two compounds with the same carboxylic acid derivative group the one with the least steric crowding on the acyl side is typically the most reactive.
b) Compound B would be expected to be the more reactive. In general acid anhydrides are more reactive than ester.
2)
a)
b)
c)

