3.13: Carboxylic Acids
- Page ID
- 107508
learning objectives
- name carboxylic acids using IUPAC (systematic) and selected common name nomenclature
- draw the structure of carboxylic acids from IUPAC (systematic) and selected common names
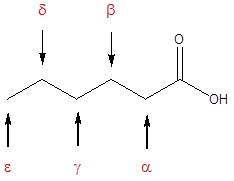
The IUPAC system of nomenclature assigns a characteristic suffix to these classes. The –e ending is removed from the name of the parent chain and is replaced -anoic acid. Since a carboxylic acid group must always lie at the end of a carbon chain, it is always is given the #1 location position in numbering and it is not necessary to include it in the name. Many carboxylic acids are called by the common names that were chosen by chemists to usually describe the origin of the compound. In common names of aldehydes, carbon atoms near the carboxyl group are often designated by Greek letters. The atom adjacent to the carbonyl function is alpha, the next removed is beta and so on.
|
Formula |
Common Name |
Source |
IUPAC Name |
Melting Point |
Boiling Point |
|---|---|---|---|---|---|
| HCO2H | formic acid | ants (L. formica) | methanoic acid | 8.4 ºC | 101 ºC |
| CH3CO2H | acetic acid | vinegar (L. acetum) | ethanoic acid | 16.6 ºC | 118 ºC |
| CH3CH2CO2H | propionic acid | milk (Gk. protus prion) | propanoic acid | -20.8 ºC | 141 ºC |
| CH3(CH2)2CO2H | butyric acid | butter (L. butyrum) | butanoic acid | -5.5 ºC | 164 ºC |
| CH3(CH2)3CO2H | valeric acid | valerian root | pentanoic acid | -34.5 ºC | 186 ºC |
| CH3(CH2)4CO2H | caproic acid | goats (L. caper) | hexanoic acid | -4.0 ºC | 205 ºC |
| CH3(CH2)5CO2H | enanthic acid | vines (Gk. oenanthe) | heptanoic acid | -7.5 ºC | 223 ºC |
| CH3(CH2)6CO2H | caprylic acid | goats (L. caper) | octanoic acid | 16.3 ºC | 239 ºC |
| CH3(CH2)7CO2H | pelargonic acid | pelargonium (an herb) | nonanoic acid | 12.0 ºC | 253 ºC |
| CH3(CH2)8CO2H | capric acid | goats (L. caper) | decanoic acid | 31.0 ºC | 219 ºC |
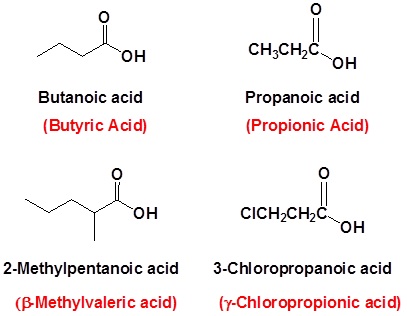
Examples (Common Names Are in Red)
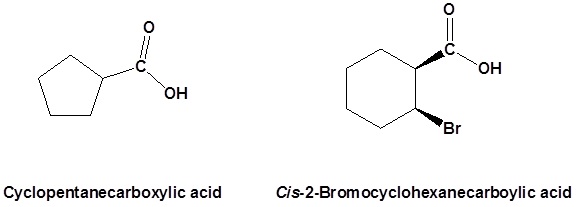
Naming carboxyl groups added to a ring
When a carboxyl group is added to a ring the suffix -carboxylic acid is added to the name of the cyclic compound. The ring carbon attached to the carboxyl group is given the #1 location number.
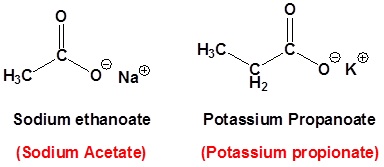
Naming carboxylates
Salts of carboxylic acids are named by writing the name of the cation followed by the name of the acid with the –ic acid ending replaced by an –ate ending. This is true for both the IUPAC and Common nomenclature systems.
Naming carboxylic acids which contain other functional groups
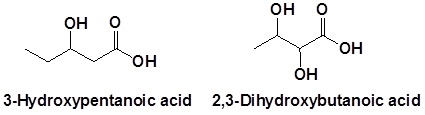
Carboxylic acids are given the highest nomenclature priority by the IUPAC system. This means that the carboxyl group is given the lowest possible location number and the appropriate nomenclature suffix is included. In the case of molecules containing carboxylic acid and alcohol functional groups the OH is named as a hydroxyl substituent. However, the l in hydroxyl is generally removed.
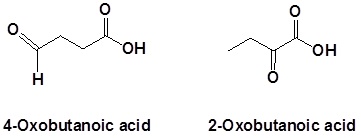
In the case of molecules containing a carboxylic acid and aldehydes and/or ketones functional groups the carbonyl is named as a "Oxo" substituent.
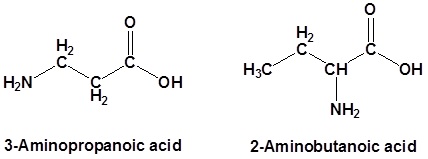
In the case of molecules containing a carboxylic acid an amine functional group the amine is named as an "amino" substituent.
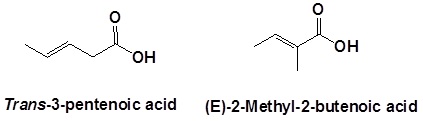
When carboxylic acids are included with an alkene the following order is followed:
(Location number of the alkene)-(Prefix name for the longest carbon chain minus the -ane ending)-(an –enoic acid ending to indicate the presence of an alkene and carboxylic acid)
Remember that the carboxylic acid has priority so it should get the lowest possible location number. Also, remember that cis/tran or E/Z nomenclature for the alkene needs to be included if necessary.
Naming dicarboxylic acids
For dicarboxylic acids the location numbers for both carboxyl groups are omitted because both functional groups are expected to occupy the ends of the parent chain. The ending –dioic acid is added to the end of the parent chain.
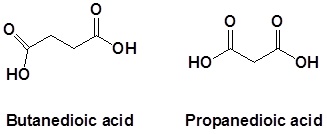
Common Names for selected dicarboxylic acids
The following common names for these selected dicarboxylic acids are important to memorize; they are prevalent in biochemistry or industrial applications.
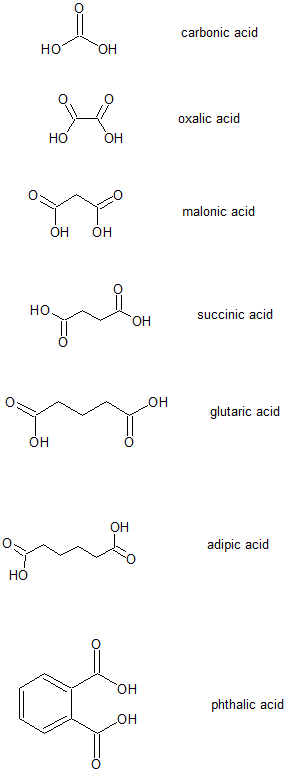
The saying, "Oh my, such good apple pie!", can help us remember these common names by correlating the first letters of each word with the common names:
oxalic acid, malonic acid, succinic acid, glutaric acid, adipic acid, and phthalic.
Exercise
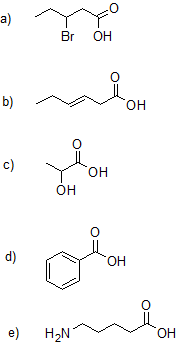
1. Draw the bond-line structure and write the condensed structural formula for each compound.
a) octanoic acid
b) 4-hydroxypentanoic acid
c) cis-4-hexenoioc acid or cis-hex-4-enoic acid
d) (E)-5-bromo-3-heptenoic acid or (E)-5-bromohept-3-enoic acid.
e) 2-aminopropanoic acid
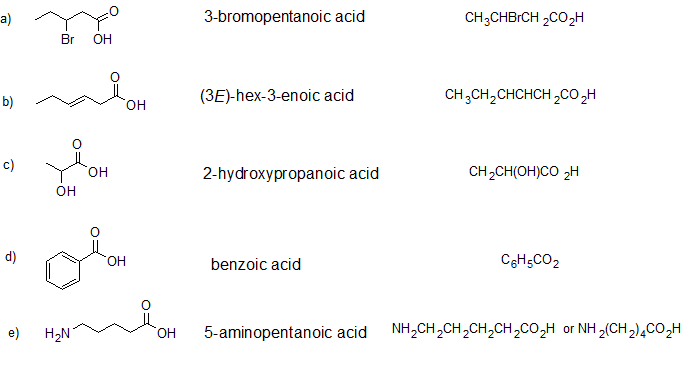
2. Give the IUPAC name and condensed structural formula for each compound.
- Answer
-
1.
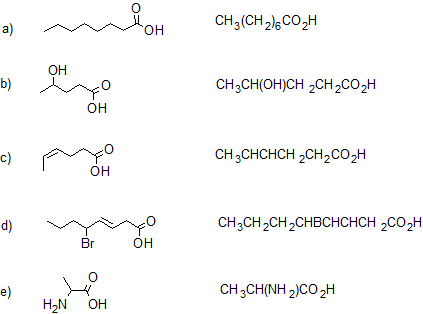
2.
Contributors and Attributions
Prof. Steven Farmer (Sonoma State University)
William Reusch, Professor Emeritus (Michigan State U.), Virtual Textbook of Organic Chemistry

