12.4: Application- Hydrogenation of Oils
- Page ID
- 30508
Introduction
In the late 1970’s the lipid hypothesis came in to existences. The lipid hypothesis states that eating saturated fats leads to elevated LDL(Low Density Lipoprotein) which is perceived to be bad cholesterol. This will result in coronary heart disease which is hardening and narrowing of arteries resulting in heart attack. Fats were eventually classified in to 2 categories, “healthy fats” and “unhealthy fats”. Unhealthy fats where perceived to be saturated fats, healthy fats where perceived to be unsaturated fats.
A meta-analysis of 72 studies with over 103,052 people have found no validity in the lipid hypothesis. The conclusion of the Meta-Analysis was,“In contrast to current recommendations, this systematic review found no evidence that saturated fat increases the risk of coronary disease, or that polyunsaturated fats have a cardio protective effect.”[1]
Dietary fats play a critical role in human health. They help keep cells healthy, help with brain development, help with the use of fat soluble vitamins, and they help cushion organs protecting them against blunt trauma. Fats come in multiple forms, Saturated, Unsaturated and trans fats just to name a few.
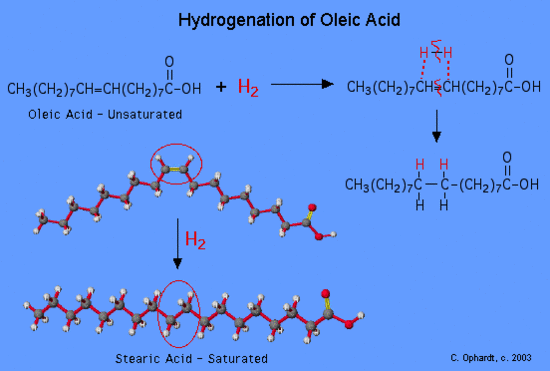
Hydrogenation Reaction
Unsaturated fatty acids may be converted to saturated fatty acids by the relatively simple hydrogenation reaction. Recall that the addition of hydrogen to an alkene (unsaturated) results in an alkane (saturated). A simple hydrogenation reaction is:
H2C=CH2 + H2 → CH3CH3
alkene plus hydrogen yields an alkane
Saturated Fats
Saturated fats are solid at room temperature due to their molecular shape. The term saturated is in reference to an Sp3 carbon chain that has its remaining Sp3 orbitals bonded with hydrogen atoms. Thus the term “saturated”. It’s “saturated” with hydrogen.
Saturated fats have a chain like structure which allows them to stack very well forming a solid at room temperature. Unsaturated fats are not linear due to double bonded carbons which results in a different molecular shape because the Sp2 carbons are trigonal planar, not tetrahedral(Sp3 carbons) as the carbons are in saturated fats. This change in structure will cause the fat molecules to not stack very well resulting in fats that are liquid at room temperature. Butter is mostly saturated fat, that’s why it’s solid at room temperature. Olive Oil is liquid at room temperature, thus it’s an unsaturated fat. An unsaturated fat can be made in to a saturated fat by a Hydrogenation process.
These are similar molecules, differing in their melting points. If the compound is a solid at room temperature, you usually call it a fat. If it is a liquid, it is often described as an oil.
Their melting points are largely determined by the presence of carbon-carbon double bonds in the molecule. The higher the number of carbon-carbon double bonds, the lower the melting point.
If there aren't any carbon-carbon double bonds, the substance is said to be saturated. A typical saturated fat might have the structure:
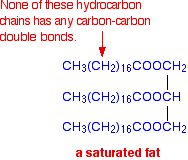
Molecules of this sort are usually solid at room temperature.
If there is only one carbon-carbon double bond in each of the hydrocarbon chains, it is called a mono-unsaturated fat (or mono-unsaturated oil, because it is likely to be a liquid at room temperature.)
A typical mono-unsaturated oil might be:
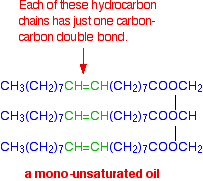
If there are two or more carbon-carbon double bonds in each chain, then it is said to be polyunsaturated.
For example:
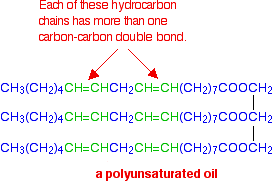
For simplicity, in all these diagrams, all three hydrocarbon chains in each molecule are the same. That doesn't have to be the case - you can have a mixture of types of chain in the same molecule.
Trans Fat
A major health concern during the hydrogenation process is the production of trans fats. Trans fats are the result of a side reaction with the catalyst of the hydrogenation process. This is the result of an unsaturated fat which is normally found as a cis isomer converts to a trans isomer of the unsaturated fat. Isomers are molecules that have the same molecular formula but are bonded together differently. Focusing on the Sp2 double bonded carbons, a cis isomer has the hydrogens on the same side. Due to the added energy from the hydrogenation process, the activation energy is reached to convert the cis isomers of the unsaturated fat to a trans isomer of the unsaturated fat. The effect is putting one of the hydrogens on the opposite side of one of the carbons. This results in a trans configuration of the double bonded carbons. The human body doesn’t recognized trans fats.
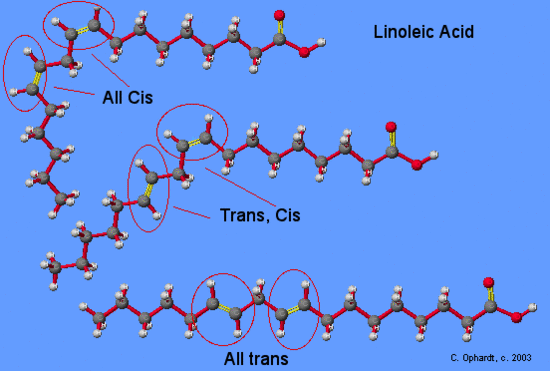
Although the trans fatty acids are chemically "monounsaturated" or "polyunsaturated" they are considered so different from the cis monounsaturated or polyunsaturated fatty acids that they can not be legally designated as unsaturated for purposes of labeling. Most of the trans fatty acids (although chemically still unsaturated) produced by the partial hydrogenation process are now classified in the same category as saturated fats.
The major negative is that trans fat tends to raise "bad" LDL- cholesterol and lower "good" HDL-cholesterol, although not as much as saturated fat. Trans fat are found in margarine, baked goods such as doughnuts and Danish pastry, deep-fried foods like fried chicken and French-fried potatoes, snack chips, imitation cheese, and confectionery fats.
Margarine manufacture
Some margarine is made by hydrogenating carbon-carbon double bonds in animal or vegetable fats and oils. You can recognise the presence of this in foods because the ingredients list will include words showing that it contains "hydrogenated vegetable oils" or "hydrogenated fats".
The impression is sometimes given that all margarine is made by hydrogenation - that's simply not true.
Vegetable oils often contain high proportions of polyunsaturated and mono-unsaturated fats (oils), and as a result are liquids at room temperature. That makes them messy to spread on your bread or toast, and inconvenient for some baking purposes.
You can "harden" (raise the melting point of) the oil by hydrogenating it in the presence of a nickel catalyst. Conditions (like the precise temperature, or the length of time the hydrogen is passed through the oil) are carefully controlled so that some, but not necessarily all, of the carbon-carbon double bonds are hydrogenated.
This produces a "partially hydrogenated oil" or "partially hydrogenated fat".
You need to hydrogenate enough of the bonds to give the final texture you want. However, there are possible health benefits in eating mono-unsaturated or polyunsaturated fats or oils rather than saturated ones - so you wouldn't want to remove all the carbon-carbon double bonds.
The downside of hydrogenation as a means of hardening fats and oils
There are some probable health risks from eating hydrogenated fats or oils. Consumers are becoming more aware of this, and manufacturers are increasingly finding alternative ways of converting oils into spreadable solids.
One of the problems arises from the hydrogenation process.
The double bonds in unsaturated fats and oils tend to have the groups around them arranged in the "cis" form.
The relatively high temperatures used in the hydrogenation process tend to flip some of the carbon-carbon double bonds into the "trans" form. If these particular bonds aren't hydrogenated during the process, they will still be present in the final margarine in molecules of trans fats.
The consumption of trans fats has been shown to increase cholesterol levels (particularly of the more harmful LDL form) - leading to an increased risk of heart disease.
Any process which tends to increase the amount of trans fat in the diet is best avoided. Read food labels, and avoid any food which contains (or is cooked in) hydrogenated oil or hydrogenated fat.
Problems
- After the hydrogenation of an unsaturated fatty acid, would it exist at room temperature as a liquid or solid?
- Write the hydrogenation reaction for linoleic acid to hydrogenate all of the double bonds. What is the new name for this fatty acid? Hint: count carbons.
- Compare a "hard" type margarine vs. a "soft" margarine. Which has the most double bonds?
- Which is the most saturated?
Contributors
- Charles Ophardt, Professor Emeritus, Elmhurst College; Virtual Chembook
- Antonio Rodriguez
Jim Clark (Chemguide.co.uk)

