24.S: Amines and Heterocycles (Summary)
- Page ID
- 207029
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \) \( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)\(\newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\) \( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\) \( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\) \( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\) \( \newcommand{\Span}{\mathrm{span}}\) \(\newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\) \( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\) \( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\) \( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\) \( \newcommand{\Span}{\mathrm{span}}\)\(\newcommand{\AA}{\unicode[.8,0]{x212B}}\)
Concepts & Vocabulary
- Design a multi‑step synthesis that involves the use of any of the reactions described in this unit.
- Solve road‑map problems that require a knowledge of amine chemistry.
- Define, and use in context, the key terms introduced.
- Amines are derivatives of ammonia in which one or more of the hydrogens has been replaced by an alkyl or aryl group.
- Several different nomenclature systems exist for naming amines, which is complicates this topic since there is no preferred option.
- The terms primary (1º), secondary (2º) & tertiary (3º) are used to classify amines by referring to the number of alkyl (or aryl) substituents bonded to the nitrogen atom.
- A nitrogen bonded to four alkyl groups will necessarily be positively charged, and is called a quaternary (4º)-ammonium cation.
- Amines are named following the IUPAC rules with -amino being added as a substitutent.
- Amines are named following the Chemical Abstract Service which the suffix -amine is attached to the root alkyl name.
- Amines are named with a common system where each alkyl substituent on nitrogen in alphabetical order, followed by the suffix -amine.
24.2 Structure and Properties of Amines
- Neutral amines have three bonds and one lone pair.
- The central nitrogen has sp3 hybridization, which leads to trigonal pyramidal geometry with bond angles of 109.5o.
- Nitrogen does not lend to isolable stereisomers since it rapidly inverts its configuration (equilibrium arrows) by passing through a planar, sp2-hybridized transition state, leading to a mixture of interconverting R and S configurations.
- Hydrogen bonding is the dominant factor for amines.
- Branching of amines (less bonds to Hydrogen) lowers the boiling point of amines.
- When evaluating the basicity of a nitrogen-containing organic functional group, the central question we need to ask ourselves is: how reactive (and thus how basic) is the lone pair on the nitrogen?
- For amines, the more alkyl groups attached, the more basic the amine is due to the electron-donating effect of the alkyl group.
- The lone pair on the nitrogen atom in an amide is in resonance with the double bond making it a more stabilized lone pair and less basic.
- When a nitrogen atom is incorporated into a ring, the bascicity depends on where the lone pair resides.
- In a ring where the lone pair on the nitrogen atom resides in a hybrid orbital, then that lone pair is basic with the more basic equating with more s character in the hybrid orbital.
- In a ring where the lone pair on the nitrogen atom resides in a p orbital, then that lone pair is delocalized and less basic.
- In extraction, one can take advantage of amines and their basicity by forming the protonated salt (RNH2+Cl−), which is soluble in water, in order to separate them.
- Amines are also acidic and the same factors that decreased the basicity of amines increase their acidity.
- Amines are often utilized as bases in reactions.
- Nitrogen atoms as part of aromatic rings are less basic than methylamine.
- The basic lone pair on the nitrogen is to some extent tied up in – and stabilized by – the aromatic p system.
- This effect is accentuated by the addition of an electron-withdrawing groups.
- Imines are somewhat basic, which can be explained using orbital theory and the inductive effect: the sp2 orbitals of an imine nitrogen are one part s and two parts p, meaning that they have about 67% s character.
24.5 Biological Amines and the Henderson-Hasselbalch Equation
- The chemistry of life occurs in a buffer that consists of a mixture of various phosphate and ammonium compounds.
- In an aqueous solution buffered at pH 7, carboxylic acid groups can be expected to be essentially 100% deprotonated and negatively charged (ie. in the carboxylate form), and amine groups essentially 100% protonated and positively charged (i.e., in the ammonium form).
- The imidizole group on the histidine side chain has a pKa near 7, and thus exists in physiological solutions as mixture of both protonated and deprotonated forms.
- Secondary amines and their salts can be synthesized from primary amines and an alkyl halide.
- Tertiary amines and their salts can be synthesized from primary amines and an alkyl halide.
- Quaternary ammonium salts can be synthesized from primary amines and an alkyl halide.
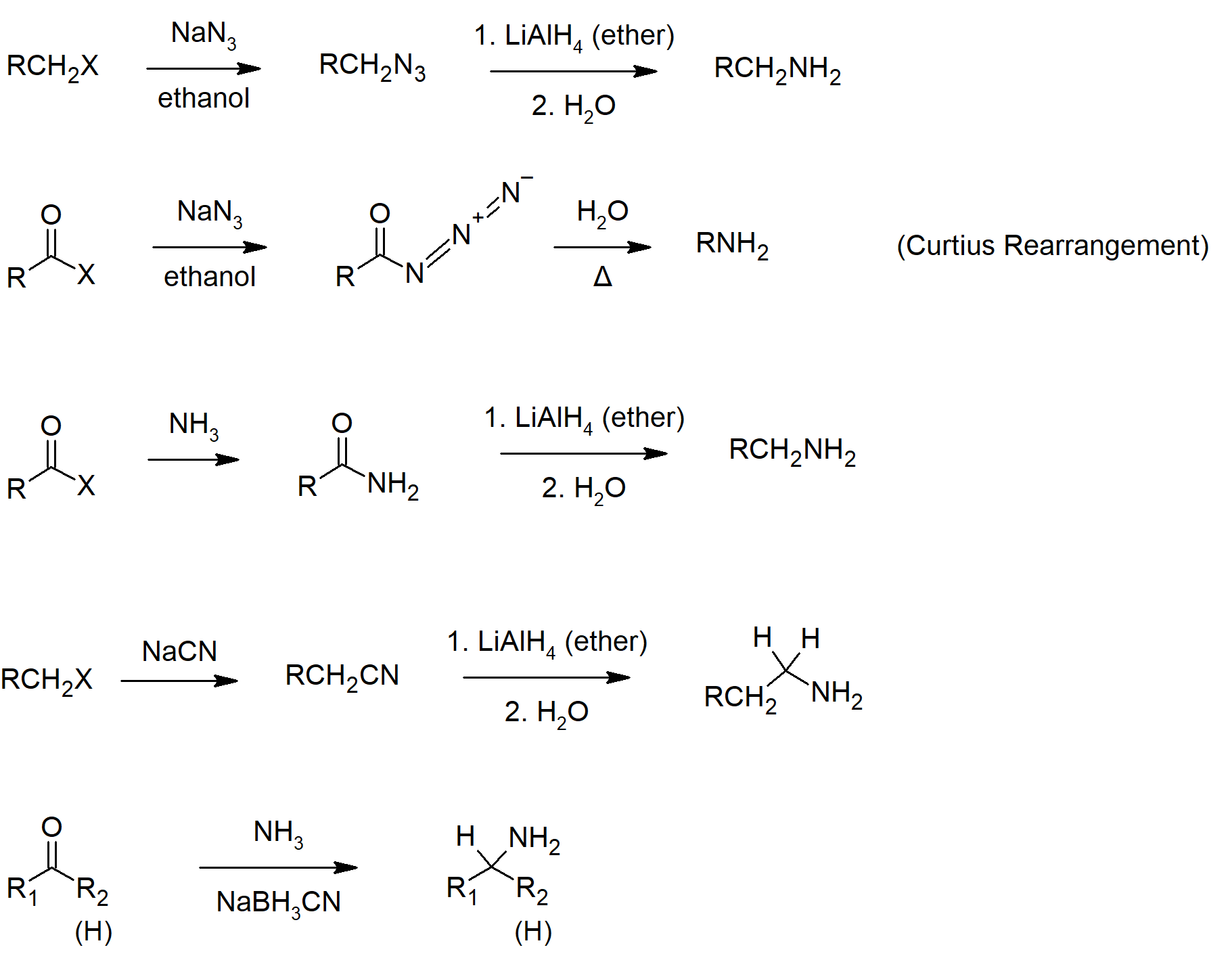
- While a primary amine can by synthesized from ammonia and an alkyl halide, there are other better options.
- The reactions that provide a more pure primary amine in high yield occur in two steps.
- First form a carbon-nitrogen bond by reacting a nitrogen nucleophile with a carbon electrophile.
- Second, any extraneous nitrogen substituents that may have facilitated this bonding are removed to give the amine product.
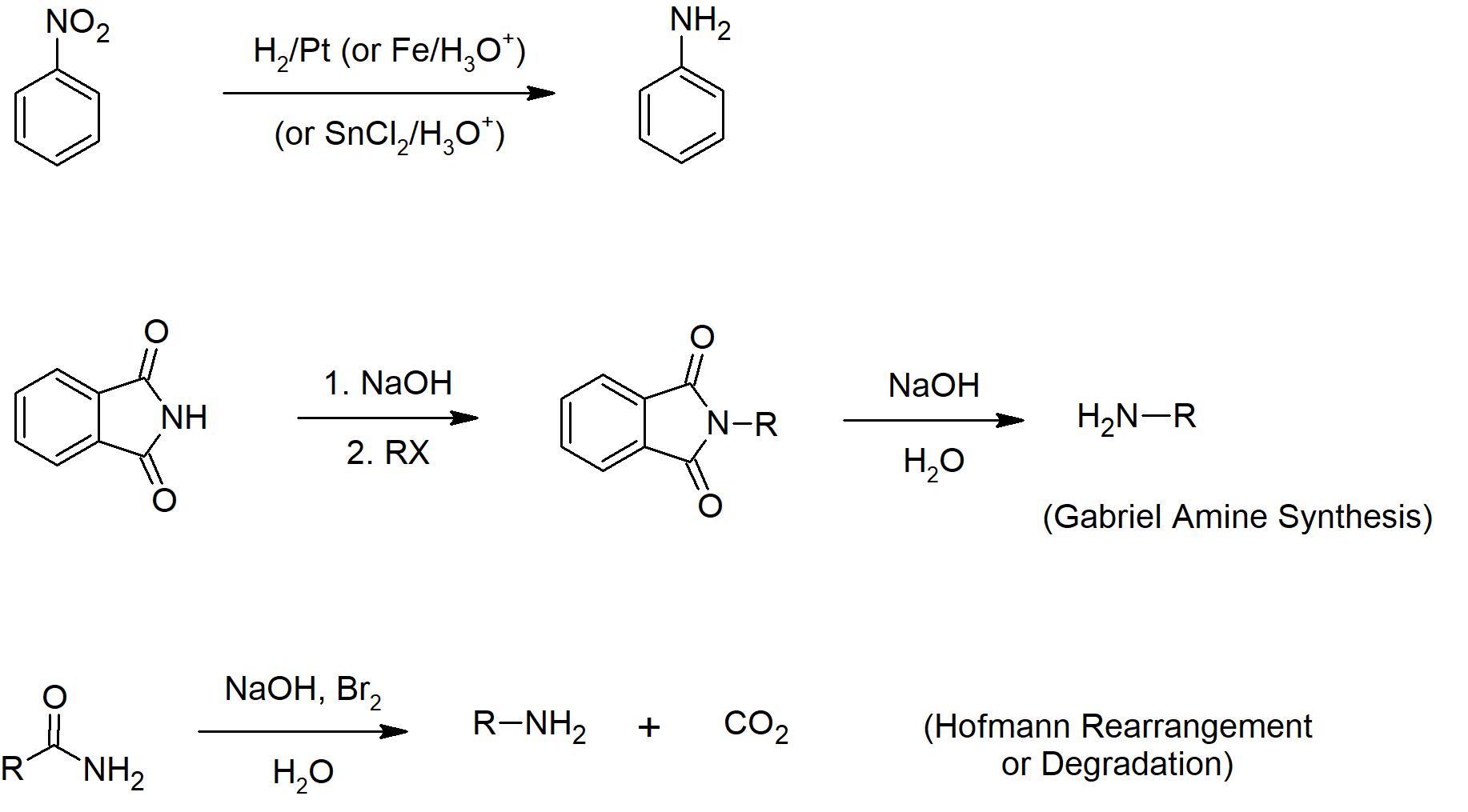
- Another option to synthesize amines is to reduce nitro groups, which include catalytic hydrogenation, zinc or tin in dilute mineral acid, and sodium sulfide in ammonium hydroxide solution.
- Reacting a nitrile with lithium aluminum hydride will yield a primary amine.
- Amides can be reduced to primary, secondary or tertiary amines using lithium aluminum hydride.
- Aldehydes and ketones can be converted to primary, secondary, or tertiary amines using reductive amination.
- Hofmann rearrangement is the reaction of a primary amide with a halogen in strongly basic conditions to give a primary amine as a product.
- The Curtius rearrangement converts an acid chloride to an amine by the migration of an -R group form the carbonyl carbon to the the neighboring nitrogen in the acyl azide intermediate.
- The direct alkylation of 1º or 2º-amines gives a more substituted product does not proceed cleanly.
- Acid chlorides can react with amines to form amides.
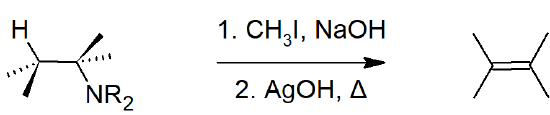
- Elimination reactions of 4º-ammonium salts are termed Hofmann eliminations, where the products are a tertiary amine and a alkene.
- The double bond Hofmann eliminations tends to give the less-substituted double bond isomer due to the Hofmann Rule.
- Arylamines are very reactive towards electrophilic aromatic substitution substitutions.
- Aniline is a strong activating group and can overreact in electrophilic aromatic substitutions.
- Changing the functional group from an amine to an amide on an aromatic ring alters the reactivity in electrophilic aromatic substitutions for more control in the reaction.
- Sulfa drugs are an important group of synthetic antimicrobial agents (pharmaceuticals) that contain the sulfonamide group.
- The synthesis of sulfanilamide (a sulfa drug) illustrates how the reactivity of aniline can be modified to make possible an electrophilic aromatic substitution.
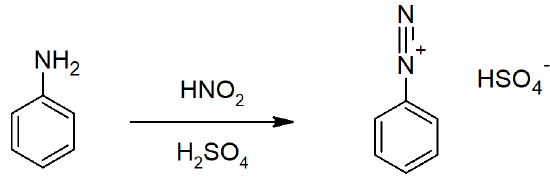
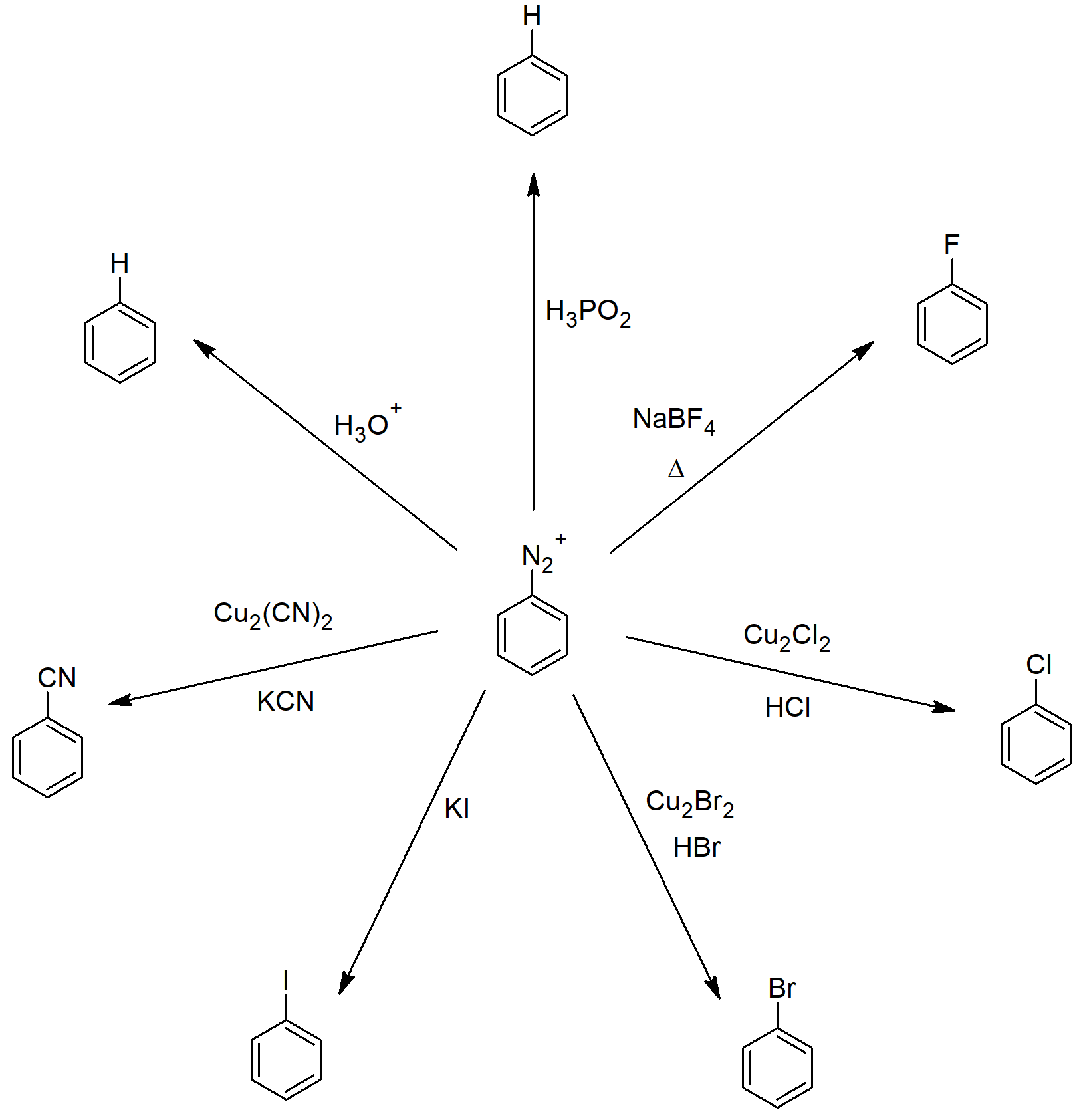
- Aryl diazonium salts are important intermediates, since the leaving group (N2) is thermodynamically very stable, these reactions are energetically favored.
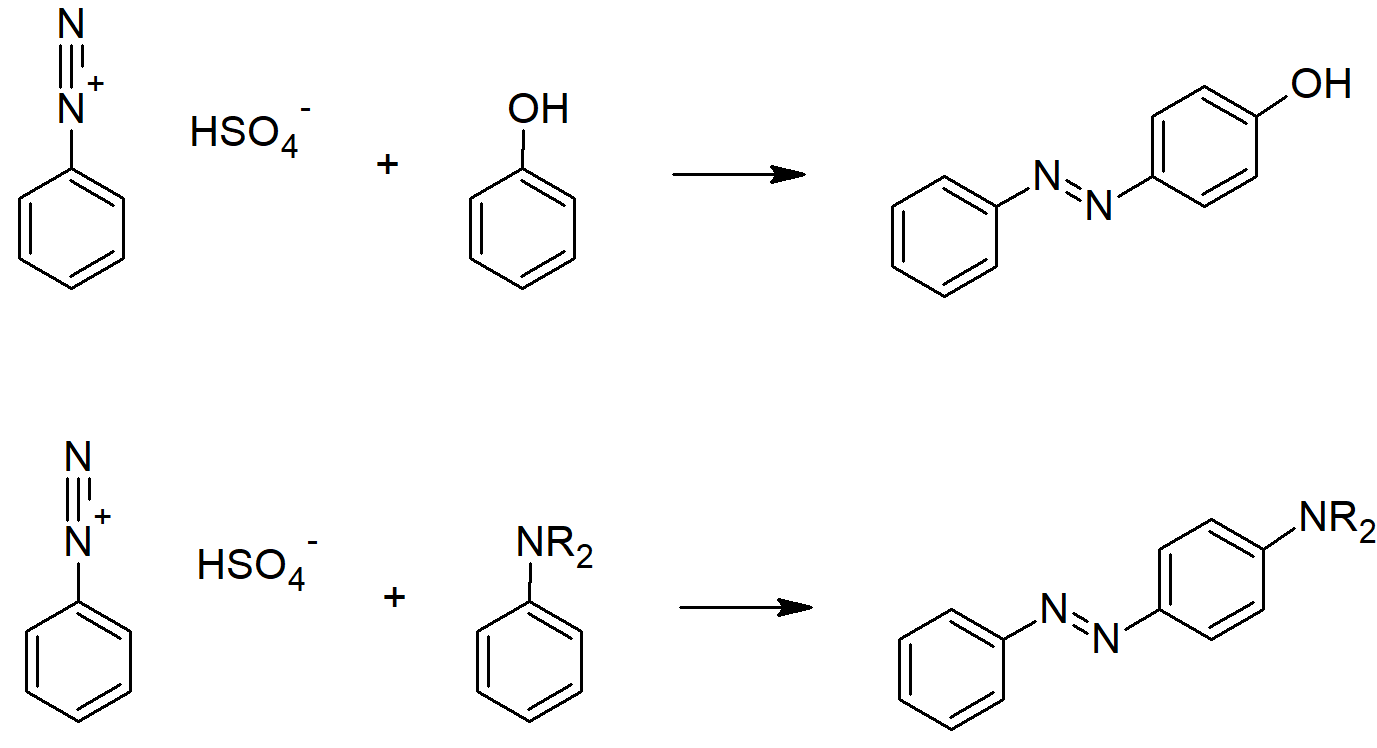
- Diazonium ions show that the positive charge is delocalized over the two nitrogen atoms while it is not possible for nucleophiles to bond to the inner nitrogen, negative nucleophiles can bond to the terminal nitrogen gives neutral azo compounds.
- The most important application of diazo coupling reactions is electrophilic aromatic substitution of activated benzene derivatives by diazonium electrophiles.
- The products of such reactions are highly colored aromatic azo compounds that find use as synthetic dyestuffs, commonly referred to as azo dyes.
- Heterocyclic structures are found in many natural products an example of some nitrogen compounds, known as alkaloids because of their basic properties.
- Pyrrole is obtained commercially by the reaction of furan with ammonia.
- In a pyrrole ring, the nitrogen lone pair is part of the aromatic sextet, thus not very basic.
- Substitution preference is on the 2-position of pyrrole.
- Imidazole is another five-membered heterocyclic amine, which is part of the amino acid histidine.
- Thiazole is a five-membered ring system which is found in biological systems.
- When a nitrogen atom is incorporated directly into an aromatic ring, its basicity depends on the bonding context.
- In pyridine, the nitrogen lone pair occupies an sp2-hybrid orbital, and is not part of the aromatic sextet, therefore, its electron pair is available for forming a bond to a proton, and thus the pyridine nitrogen atom is somewhat basic.
- The aromatic stabilization energy of pyridine is 21 kcal/mole based on heat of combustion measurements.
- Polycyclic heterocyclic structures are found in many natural products like caffeine.
- Derivatives of the simple fused ring heterocycle purine constitute an especially important and abundant family of natural products.
- The amino compounds adenine and guanine are two of the complementary bases that are essential components of DNA.
- The hydrogens attached to an amine show up ~ 0.5-5.0 ppm in 1H NMR.
- The broad range is due to the fact that the location is dependent on the amount of hydrogen bonding and the sample's concentration.
- The hydrogens on carbons directly bonded to an amine typically appear ~2.3-3.0 ppm in 1H NMR.
- IR for primary amines a free N-H absorption is observed in the 3400 to 3500 cm-1 region as two well-defined peaks.
- IR for amines the C-N stretching absorptions are found at 1200 to 1350 cm-1 for aromatic amines, and at 1000 to 1250 cm-1 for aliphatic amines.
- Secondary amines exhibit only one absorption near 3420 cm-1 in IR.
- For tertiary amines, there is not N-H stretch.
Skills to Master
- Skill 24.1 Name amines using IUPAC rules.
- Skill 24.2 Draw the structure of amines from the IUPAC name.
- Skill 24.3 Describe the geometries and approximate bond angles of amines.
- Skill 24.4 Explain physical properties of amines.
- Skill 24.5 Explain why one amine is more basic than another.
- Skill 24.6 Explain how the basicity of an amine when the Nitrogen atom is incorporated in a ring depends on the bonding context.
- Skill 24.7 Describe how an amine can be extracted from a mixture that also contains neutral compounds illustrating the reactions which take place with appropriate equations.
- Skill 24.8 Explain why primary and secondary (but not tertiary) amines may be regarded as very weak acids.
- Skill 24.9 Use the concept of resonance to explain why arylamines are less basic than their aliphatic counterparts.
- Skill 24.10 Arrange a given series of arylamines in order of increasing or decreasing basicity.
- Skill 24.11 Explain using inductive and resonance effects arguments as to why a given arylamine is more or less basic than aniline.
- Skill 24.12 Identify the form that amine bases take within living cells.
- Skill 24.13 Use the Henderson‑Hasselbalch equation to calculate the percentage of a base that is protonated in a solution, given the pKa value of the associated ion and the pH of the solution.
- Skill 24.14 List general ways to synthesize amines.
- Skill 24.15 Draw mechanisms for preparing amines including:
- reduction of nitriles, amides and nitro compounds.
- reactions involving alkyl groups:
- SN2 reactions of alkyl halides, ammonia and other amines.
- nucleophilic attack by an azide ion on an alkyl halide, followed by reduction of the azide so formed.
- alkylation of potassium phthalimide, followed by hydrolysis of the N‑alkyl phthalimide so formed (i.e., the Gabriel synthesis).
- reductive amination of aldehydes or ketones.
- Hofmann or Curtius rearrangements.
- Skill 24.16 Write an equation to represent the reaction that takes place between ammonia, a primary or secondary amine, and an acid chloride.
- Skill 24.17 Identify the product formed when a given amine reacts with a given acid chloride.
- Skill 24.18 Draw the product for a Hoffman elimination.
- Skill 24.19 Propose a synthesis of arylamines using diazonium coupling reactions.
- Skill 24.20 Be able to draw the structure of furan, pyrrole and imidazole.
- Skill 24.21 Use the Hückel 2n + 4 rule to explain the aromaticity of pyrrole.
- Skill 24.22 Be able to predict the product formed when pyrrole is subjected to an aromatic electrophilic substitution.
- Skill 24.23 Write the detailed mechanism for the electrophilic aromatic substitution of pyrrole to account for the fact that substitution takes place at C2 rather than C3.
- Skill 24.24 Explain the difference in basicity between pyridine, pyrrole and other amines.
- Skill 24.25 Explain why pyridine undergoes electrophilic substitution much less readily than does benzene.
- Skill 24.17 Use IR, NMR and MS in combination with the nitrogen rule to identify amines.
Summary of Reactions
Amine Preparation

Amine Reactions