22.18: Some Naturally Occurring Products Derived from Carbohydrates
- Page ID
- 16504
Cotton, probably the most useful natural fiber, is nearly pure cellulose. The manufacture of textiles from cotton involves physical manipulation of the raw material by carding, combing and spinning selected fibers. For fabrics the best cotton has long fibers, and short fibers or cotton dust are removed. Crude cellulose is also available from wood pulp by dissolving the lignan matrix surrounding it. These less desirable cellulose sources are widely used for making paper.
In order to expand the ways in which cellulose can be put to practical use, chemists have devised techniques for preparing solutions of cellulose derivatives that can be spun into fibers, spread into a film or cast in various solid forms. A key factor in these transformations are the three free hydroxyl groups on each glucose unit in the cellulose chain, --[C6H7O(OH)3]n--. Esterification of these functions leads to polymeric products having very different properties compared with cellulose itself.
- Cellulose Nitrate, first prepared over 150 years ago by treating cellulose with nitric acid, is the earliest synthetic polymer to see general use. The fully nitrated compound, --[C6H7O(ONO2)3]n--, called guncotton, is explosively flammable and is a component of smokeless powder. Partially nitrated cellulose is called pyroxylin. Pyroxylin is soluble in ether and at one time was used for photographic film and lacquers. The high flammability of pyroxylin caused many tragic cinema fires during its period of use. Furthermore, slow hydrolysis of pyroxylin yields nitric acid, a process that contributes to the deterioration of early motion picture films in storage.
- Cellulose Acetate, --[C6H7O(OAc)3]n--, is less flammable than pyroxylin, and has replaced it in most applications. It is prepared by reaction of cellulose with acetic anhydride and an acid catalyst. The properties of the product vary with the degree of acetylation. Some chain shortening occurs unavoidably in the preparations. An acetone solution of cellulose acetate may be forced through a spinneret to generate filaments, called acetate rayon, that can be woven into fabrics.
- Viscose Rayon, is prepared by formation of an alkali soluble xanthate derivative that can be spun into a fiber that reforms the cellulose polymer by acid quenching. The following general equation illustrates these transformations. The product fiber is called viscose rayon.
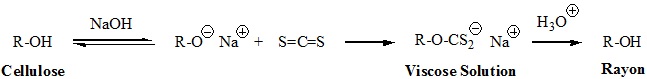
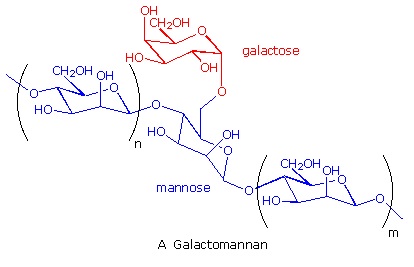
Many complex polysaccharides are found in nature. The galactomannans, consisting of a mannose backbone with galactose side groups, are an interesting and useful example. A (1-4)-linked beta-D-mannose chain is adorned with 1-6-linked alpha-D-galactose units, as shown in the diagram below. The ratio of galactose to mannose usually ranges from 1:2 to 1:4. An important source of this substance is the guar bean, grown principally in northwestern India, and Pakistan. Altough guar protein is not of nutritional value to humans (guar means 'cow food' in Hindi), the bean is important as a source of guar gum, a galactomannan which forms a gel in water. The food industry uses this material as a stabilizer in ice cream, cream cheese and salad dressings.
Recently, guar gum has becomes an essential ingredient for mining oil and natural gas in a process called hydraulic fracturing. The demand for guar has increased to such a degree that the poor farmers in northwestern India have had their lives transformed by unexpected wealth.
Contributors
Prof. Steven Farmer (Sonoma State University)
William Reusch, Professor Emeritus (Michigan State U.), Virtual Textbook of Organic Chemistry

