10.2: Other Methods Used to Convert Alcohols into Alkyl Halide
- Page ID
- 13898
This page looks at reactions in which the -OH group in an alcohol is replaced by a halogen such as chlorine or bromine. It includes a simple test for an -OH group using phosphorus(V) chloride. The general reaction looks like this:
\[ ROH + HX \rightarrow RX + H_2O \]
Reaction with hydrogen chloride
Tertiary alcohols react reasonably rapidly with concentrated hydrochloric acid, but for primary or secondary alcohols the reaction rates are too slow for the reaction to be of much importance. A tertiary alcohol reacts if it is shaken with with concentrated hydrochloric acid at room temperature. A tertiary halogenoalkane (haloalkane or alkyl halide) is formed.

Replacing -OH by bromine
Rather than using hydrobromic acid,the alcohol is typically treated with a mixture of sodium or potassium bromide and concentrated sulfuric acid. This produces hydrogen bromide, which reacts with the alcohol. The mixture is warmed to distil off the bromoalkane.
\[ CH_3CH_2OH + HBr \rightarrow CH_3CH_2Br + H_2O \label{1.1.2}\]
Replacing -OH by iodine
In this case, the alcohol is reacted with a mixture of sodium or potassium iodide and concentrated phosphoric(V) acid, H3PO4, and the iodoalkane is distilled off. The mixture of the iodide and phosphoric(V) acid produces hydrogen iodide, which reacts with the alcohol.
\[ CH_3CH_2OH + HI \rightarrow CH_3CH_2I + H_2O \label{1.1.3}\]
Phosphoric(V) acid is used instead of concentrated sulfuric acid because sulfuric acid oxidizes iodide ions to iodine and produces hardly any hydrogen iodide. A similar phenomenon occurs to some extent with bromide ions in the preparation of bromoalkanes but not enough to interfere with the main reaction. There is no reason why you could not use phosphoric(V) acid in the bromide case instead of sulfuric acid if desired.
Reacting Alcohols with Phosphorus Halides
Alcohols react with liquid phosphorus(III) chloride (also called phosphorus trichloride) to yield chloroalkanes.
\[ 3CH_3CH_2CH_2OH + PCl_3 \rightarrow 3CH_3CH_2CH_2Cl + H_3PO_3 \label{1.1.3a}\]
Alcohols also violently react with solid phosphorus(V) chloride (phosphorus pentachloride) at room temperature, producing clouds of hydrogen chloride gas. While it is not a good approach to make chloroalkanes, it is a good test for the presence of -OH groups. To show that a substance was an alcohol, you would first have to eliminate all the other groups that also react with phosphorus(V) chloride. For example, carboxylic acids (containing the -COOH group) also react with it (because of the -OH in -COOH) as does water (H-OH).
If you have a neutral liquid not contaminated with water, and clouds of hydrogen chloride are produced when you add phosphorus(V) chloride, then you have an alcohol group present.
\[ CH_3CH_2CH_2OH + PCl_5 \rightarrow CH_3CH_2CH_2Cl + POCl_3 + HCl \label{1.1.4}\]
There are also side reactions involving the \(POCl_3\) reacting with the alcohol.
Other reactions involving phosphorus halides
Instead of using phosphorus(III) bromide or iodide, the alcohol is usually heated under reflux with a mixture of red phosphorus and either bromine or iodine. The phosphorus first reacts with the bromine or iodine to give the phosphorus(III) halide.
\[ 2P_{(s)} + 3Br_2 \rightarrow 2PBr_3\label{1.1.5}\]
\[ 2P_{(s)} + 3I_2 \rightarrow 2PI_3 \label{1.1.6}\]
These then react with the alcohol to give the corresponding halogenoalkane, which can be distilled off.
\[ 3CH_3CH_2CH_2OH + PBr_3 \rightarrow 3CH_3CH_2CH_2Br + H_3PO_3 \label{1.1.7}\]
\[ 3CH_3CH_2CH_2OH + PI_3 \rightarrow 3CH_3CH_2CH_2I + H_3PO_3 \label{1.1.8}\]
Reacting alcohols with Thionyl Chloride
Sulfur dichloride oxide (thionyl chloride) has the formula SOCl2. Traditionally, the formula is written as shown, despite the fact that the modern name writes the chlorine before the oxygen (alphabetical order). The sulfur dichloride oxide reacts with alcohols at room temperature to produce a chloroalkane. Sulfur dioxide and hydrogen chloride are given off. Care would have to be taken because both of these are poisonous.
\[ CH_3CH_2CH_2OH + SOCl_2 \rightarrow CH_3CH_2CH_2Cl + SO_2 + HCl \label{1.1.9}\]
The advantage that this reaction has over the use of either of the phosphorus chlorides is that the two other products of the reaction (sulfur dioxide and HCl) are both gases. That means that they separate themselves from the reaction mixture.
Contributors
Jim Clark (Chemguide.co.uk)
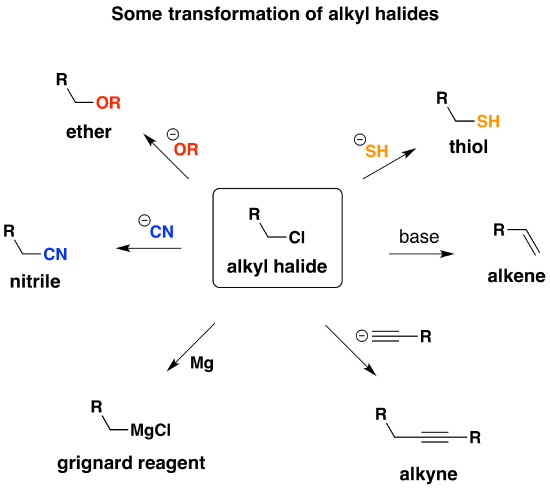
If There is one thing you learn how to do well in Org 1, it’s make alcohols. Let’s count the ways: hydroboration, acid-catalyzed hydration, oxymercuration for starters, and then substitution of alkyl halides with water or \(HO^-\). If you want to extend it even further, There is dihydroxylation (to make diols) using \(OsO_4\) or cold \(KMnO_4\), and even opening of epoxides under acidic or basic conditions to give alcohols.
There is just one issue here and it comes up once you try to use alcohols in synthesis. Let’s say you want to use that alcohol in a subsequent substitution step, getting rid of the \(HO^-)\ and replacing it with something else. See any problems with that? Remember that good leaving groups are weak bases – and the hydroxide ion, being a strong base, tends to be a pretty bad leaving group.
So what can we do?
What you want to do is convert the alcohol into a better leaving group. One way is to convert the alcohol into a sulfonate ester – we talked about that with \( TsCl\) and \(MsCl\). Alternatively, alcohols can be converted into alkyl chlorides with thionyl chloride (\(SOCl_2\)). This is a useful reaction, because the resulting alkyl halides are versatile compounds that can be converted into many compounds that are not directly accessible from the alcohol itself.
If you take an alcohol and add thionyl chloride, it will be converted into an alkyl chloride. The byproducts here are hydrochloric acid (\(HCl\)) and sulfur dioxide (\(SO_2\)). Note: there are significant differences in how this reaction is taught at different schools. Consult your instructor to be 100% sure that this applies to your course). See post here
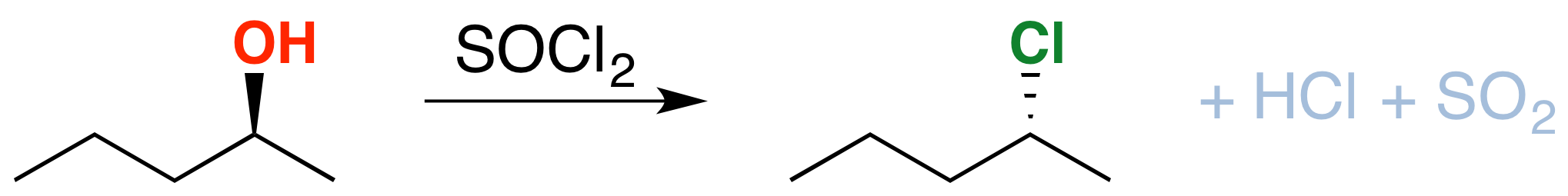
There is one important thing to note here: see the stereochemistry? It’s been inverted.*(white lie alert – see below) That’s an important difference between \(SOCl_2\) and TsCl, which leaves the stereochemistry alone. We’ll get to the root cause of that in a moment, but in the meantime, can you think of a mechanism which results in inversion of configuration at carbon?
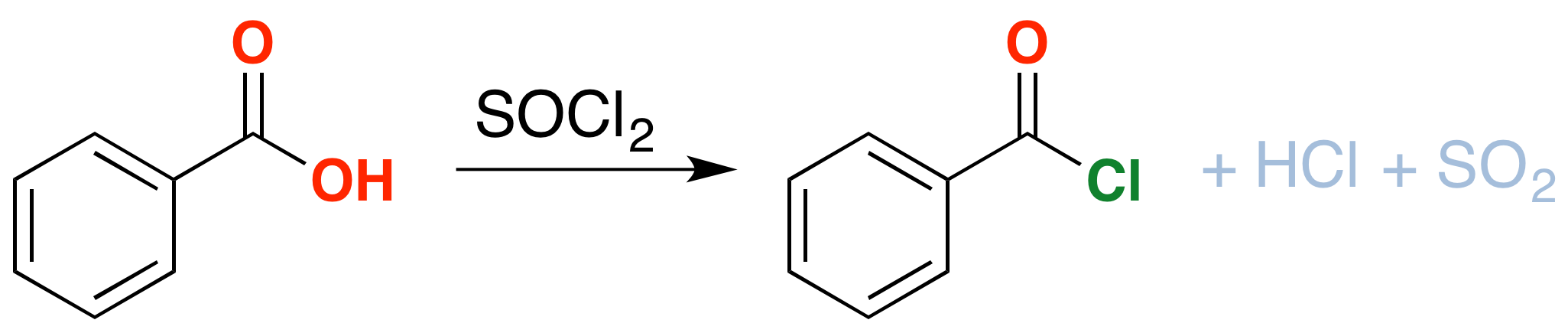
As an extra bonus, thionyl chloride will also convert carboxylic acids into acid chlorides (“acyl chlorides”). Like alcohols, carboxylic acids have their limitations as reactants: the hydroxyl group interferes with many of the reactions we learn for nucleophilic acyl substitution (among others). Conversion of the OH into Cl solves this problem.
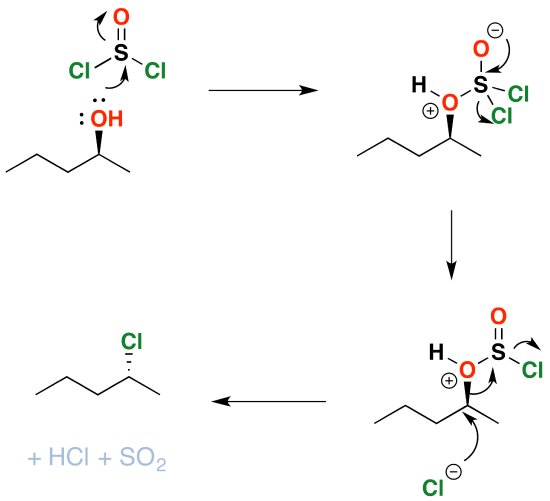
Mechanism
As you might have guessed, conversion of alcohols to alkyl halides proceeds through a substitution reaction – specifically, an \(S_N2\) mechanism. The first step is attack of the oxygen upon the sulfur of \(SOCl_2\), which results in displacement of chloride ion. This has the side benefit of converting the alcohol into a good leaving group: in the next step, chloride ion attacks the carbon in \(S_N2\) fashion, resulting in cleavage of the C–O bond with inversion of configuration. The \(HOSCl\) breaks down into \(HCl\) and sulfur dioxide gas, which bubbles away.
Formation of Alkyl Chlorides
Since the reaction proceeds through a backside attack (\(S_N2\)), there is inversion of configuration at the carbon
The mechanism for formation of acid chlorides from carboxylic acids is similar. The conversion of caboxylic acids to acid chlorides is similar, but proceeds through a [1,2]-addition of chloride ion to the carbonyl carbon followed by [1,2]-elimination to give the acid chloride, \(SO_2\) and \(HCl\)
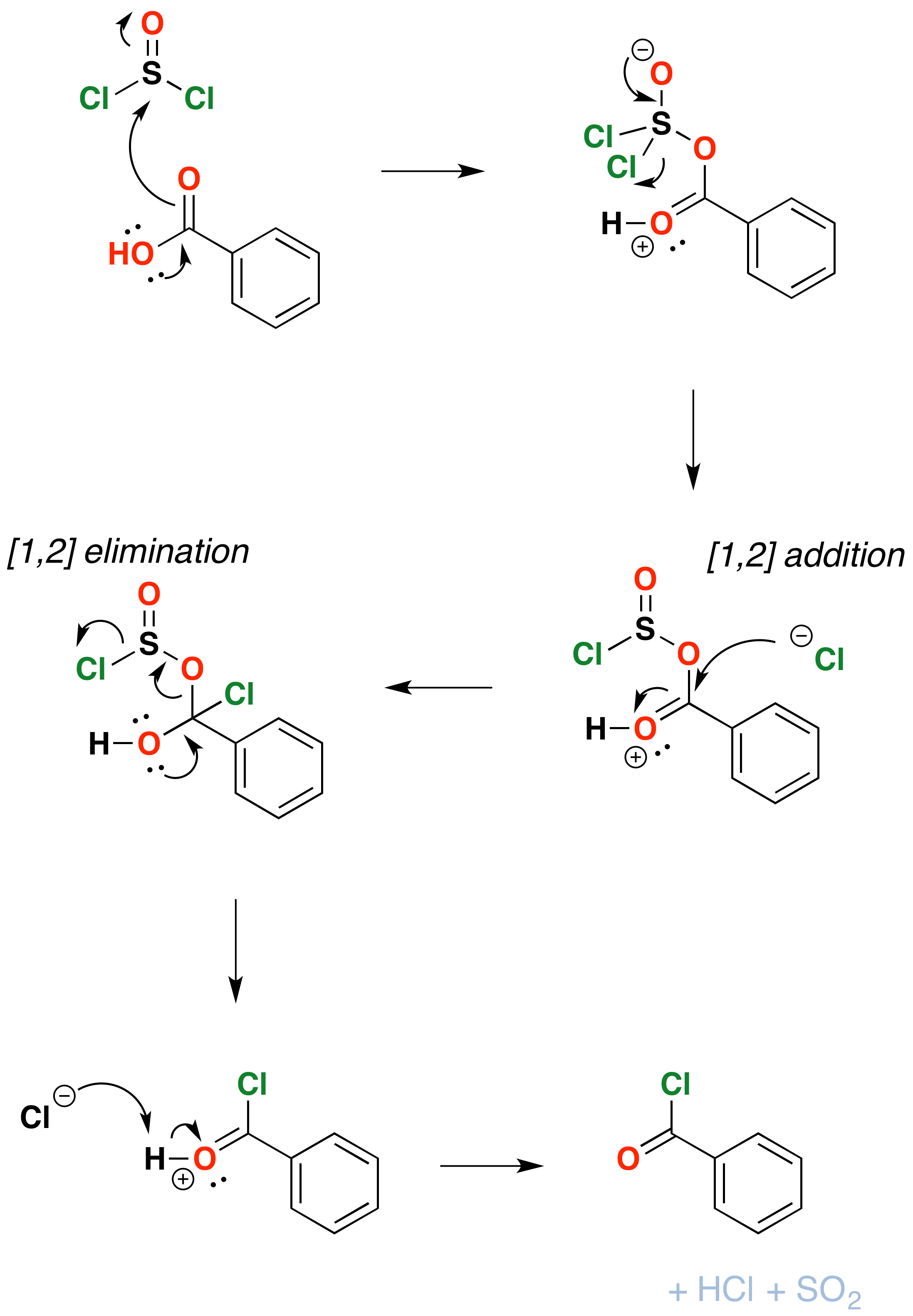
In the laboratory
Like many sulfur-containing compounds, thionyl chloride is noseworthy for its pungent smell. Thionyl chloride has a nauseating sickly-sweet odor to it that imprints itself forever upon your memory. One accident that occurred during my time as a TA involved a student dropping a flask with 5 mL of thionyl chloride into a rotovap bath outside the fume hood. The cloud of \(SO_2\) and \(HCl\) that formed cleared the teaching lab for half an hour, so you can imagine what thionyl chloride would do if exposed to the moisture in your lungs. Treat with caution, just as you would if you were working with phosgene.
*Here’s the white lie. Although it’s often taught that \(SOCl_2\) leads to 100% inversion of configuration, in reality it’s not always that simple. Inversion of configuration with \(SOCl_2\) is very solvent dependent. Depending on the choice of solvent, one can get either straight inversion, or a mixture of retention and inversion. For the purposes of beginning organic classes, most students can ignore this message.
Contributors
James Ashenhurst (MasterOrganicChemistry.com)

