9.7: Stereochemistry of Reactions - Hydration of Achiral Alkenes
- Page ID
- 110174
Learning Objective
- discern the stereochemical differences between the EAR of chiral and achiral alkenes
Stereochemistry and the Subtle Details
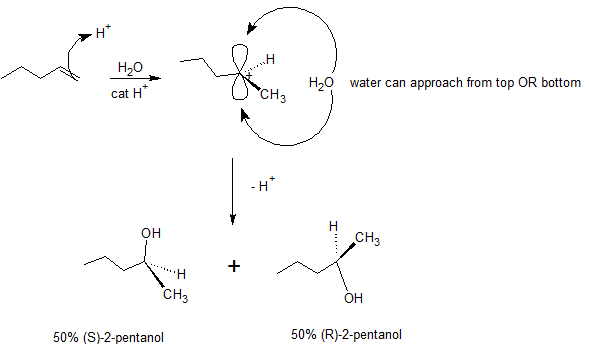
Organic reactions in the laboratory or in living systems can produce chiral centres. Consider reaction of 1-pentene with water (acid catalyzed). Markovnikov regiochemistry occurs and the OH adds to the second carbon. However, both R and S products occur giving a racemic (50/50) mixture of 2‑butanol. How does this occur? The proton addition to 1‑pentene results in a planar carbocation intermediate. A molecule of water is then equally likely to react from the top or the bottom of this cation to produce either (S)‑2‑pentanol or (R)‑2‑pentanol, respectively, as shown in the mechanism below.
Contributors and Attributions
Dr. Dietmar Kennepohl FCIC (Professor of Chemistry, Athabasca University)
Prof. Steven Farmer (Sonoma State University)