30.9: Rearrangements of Acyl Carbenes
- Page ID
- 44008
1. The Arndt-Eistert Reaction
The rearrangement of acyl nitrenes to isocyanates that is the crux of the Hofmann, Curtius and Lossen rearrangements, is paralleled by the rearrangement of acyl carbenes to ketenes, a transformation called the Wolff rearrangement. This rearrangement is a critical step in the Arndt-Eistert procedure for elongating a carboxylic acid by a single methylene unit, as described in the diagram below. The starting acid, written on the left, is converted first to an acyl chloride derivative, and then to a diazomethyl ketone. Diazomethane has a nucleophilic methylene group, as indicated by the resonance formulas drawn in the shaded box. Acylation of the methylene carbon produces an equilibrium mixture of a diazonium species and the diazomethyl ketone plus hydrogen chloride (written in brackets). If the HCl is not neutralized by a base, this mixture reacts further to give a chloromethyl ketone with loss of nitrogen. However, if the HCl is neutralized as it is formed, the relatively stable diazo ketone is obtained and may be used in subsequent reactions.
Since diazomethane itself may function as a base, the course of a given reaction is established by the manner in which the reactants are combined. When an ether solution of diazomethane is slowly added to a warm solution of the acid chloride, nitrogen evolution is observed and the chloromethyl ketone is the chief product. One equivalent of diazomethane is required for this reaction. If the addition is reversed, so that a cold solution of the acid chloride is added slowly to an excess of diazomethane in cold ether solution, nitrogen evolution is again observed; but two equivalents of diazomethane are consumed. The products are the diazo ketone and methyl chloride (a gas) from the reaction of diazomethane with HCl..
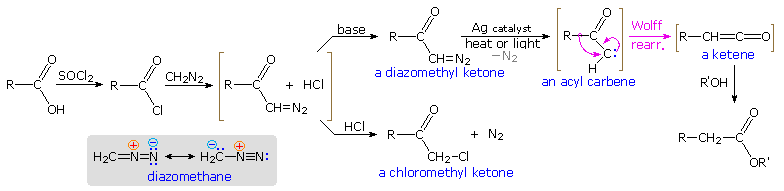
To carry out the Arndt-Eistert reaction the diazo ketone is decomposed in the presence of a silver catalyst (usually AgO2 or AgNO3 ) together with heat or light energy. The resulting Wolff rearrangement generates a ketene, which quickly reacts with any hydroxylic or amine reactants that may be present in solution. The general equations on the left below illustrate that the end product from the Arndt-Eistert reaction may be a carboxylic acid, an ester or an amide.
| 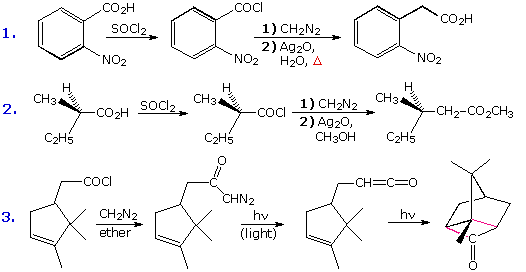 |
Three specific examples of this procedure are presented to the right of the general equations. The first two examples are typical Arndt-Eistert reactions. Tolerance for other functional groups, such as nitro, is demonstrated by the first case; and the second example shows that the configuration of the migrating group is retained in the rearrangement. Other ketene reactions, such as a [2 + 2] cycloaddition, may take place in the absence of hydroxylic solvents or amines. Reaction #3 is an example of such an alternative reaction. The new bonds in the cycloadduct are colored pink.
2. Diazo Ketone Reactions
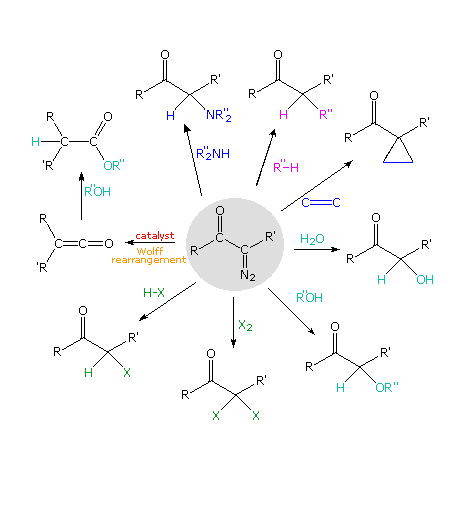
The Arndt-Eistert reaction is a special case of a more general class of diazo ketone reactions. If we assume that diazo ketones normally decompose to acyl carbenes, then numerous subsequent reactions can be imagined, and many have been realized. The following diagram outlines some of these transformations, originating from the diazo ketone formula in the center of the diagram. Molecular nitrogen is lost in each case, and the Wolff rearrangement path is on the left. Most of the other reactions reflect the ability of carbenes to insert into sigma bonds or add to double bonds. Metal catalysts are sometimes used to facilitate certain reactions, and may associate with carbenes to generate carbenoid intermediates.
By clicking on the diagram some examples of these diazoketone reactions will be shown.


 RCH2CO2H
RCH2CO2H